Which of the following are isomers?
A) ethane and propane
B) ethane and ethene
C) ethane and ethyne
D) butane and isobutane
Answer
610.5k+ views
Hint: These are the examples of structural isomers. One of the compounds has a molecular formula as \[C{H_3}C{H_2}C{H_2}C{H_3}\]. One compound is branched, while the other one is unbranched.
Complete answer:
Butane and isobutane are isomers. To be precise, these are the examples of structural isomers.
Now, come to the point of what this isomer is. Isomers are the molecules, which have identical molecular formulas, which means they have the same number of atoms for each element in a compound. But the arrangements of the atoms in space are different from each other. The isomers do not necessarily exhibit the similarity in chemical or physical properties.
The isomer is of two types. One is the structural or constitutional isomer and the other is the stereoisomer or spatial isomer.
Here, butane and isobutane are structural isomers. In structural isomerism, molecules have the same number of atoms of each element but the bonds between the atoms differ.
The structure of butane and isobutane are shown below:
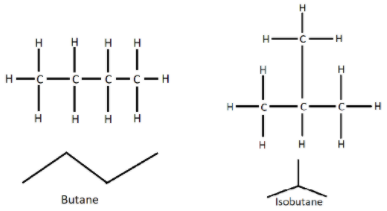
So, The correct answer is (D).
Additional Information: The main difference between butane and isobutane is that isobutane is the structural isomer of butane. Butane can convert into isobutane through the isobutane production process named isomerization. Isobutane also is a flammable hydrocarbon gas, which can be liquified through pressurization. But isobutane has different physical properties. For example, isobutane is a colorless gas with a petrol odour.
Note:
So, we can see that the molecular formula of butane is \[C{H_3}C{H_2}C{H_2}C{H_3}\]. It is an unbranched alkane molecule having four carbon atoms. On the other hand, isobutane or i-butane has the molecular formula of \[HC{\left( {C{H_3}} \right)_3}\], which is the simplest alkane with tertiary carbon, which means it has a branched structure. So, it is a structural isomer of butane.
Complete answer:
Butane and isobutane are isomers. To be precise, these are the examples of structural isomers.
Now, come to the point of what this isomer is. Isomers are the molecules, which have identical molecular formulas, which means they have the same number of atoms for each element in a compound. But the arrangements of the atoms in space are different from each other. The isomers do not necessarily exhibit the similarity in chemical or physical properties.
The isomer is of two types. One is the structural or constitutional isomer and the other is the stereoisomer or spatial isomer.
Here, butane and isobutane are structural isomers. In structural isomerism, molecules have the same number of atoms of each element but the bonds between the atoms differ.
The structure of butane and isobutane are shown below:

So, The correct answer is (D).
Additional Information: The main difference between butane and isobutane is that isobutane is the structural isomer of butane. Butane can convert into isobutane through the isobutane production process named isomerization. Isobutane also is a flammable hydrocarbon gas, which can be liquified through pressurization. But isobutane has different physical properties. For example, isobutane is a colorless gas with a petrol odour.
Note:
So, we can see that the molecular formula of butane is \[C{H_3}C{H_2}C{H_2}C{H_3}\]. It is an unbranched alkane molecule having four carbon atoms. On the other hand, isobutane or i-butane has the molecular formula of \[HC{\left( {C{H_3}} \right)_3}\], which is the simplest alkane with tertiary carbon, which means it has a branched structure. So, it is a structural isomer of butane.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




