Which is the correct statement about $C{{r}_{2}}O_{7}^{2-}$ structure?
A.It has neither $Cr-Cr$ bonds nor $O-O$ bonds.
B.It has one $Cr-Cr$ bond and six $O-O$ bonds.
C.It has no $Cr-Cr$ bonds and six $O-O$ bonds.
D.It has one $Cr-Cr$ bond and seven $Cr-O$ bonds.
Answer
543.9k+ views
Hint: We know that the Dichromate ion $\left( C{{r}_{2}}O_{7}^{2-} \right)$ is an anionic part of its salt. Dichromate ion is one of the divalent inorganic anions. These are oxyanions having \[+6\] oxidation state of Chromium. These are very good oxidizing agents due to the attachment of oxygen atoms.
Complete answer:
As we know the concept of chemical bonding and structures of ions. Also, the bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule. Bond lengths are expressed in terms of Angstrom or picometer. The two $Cr-O$ bonds which share the single oxygen atom are longer than the six $Cr-O$ terminal bonds that are equivalent. The salt of the dichromate ion is used for oxidation and reduction reactions. Dichromate poses an orange colour. The dichromate dianion consists of two tetrahedra sharing oxygen at the common corner.
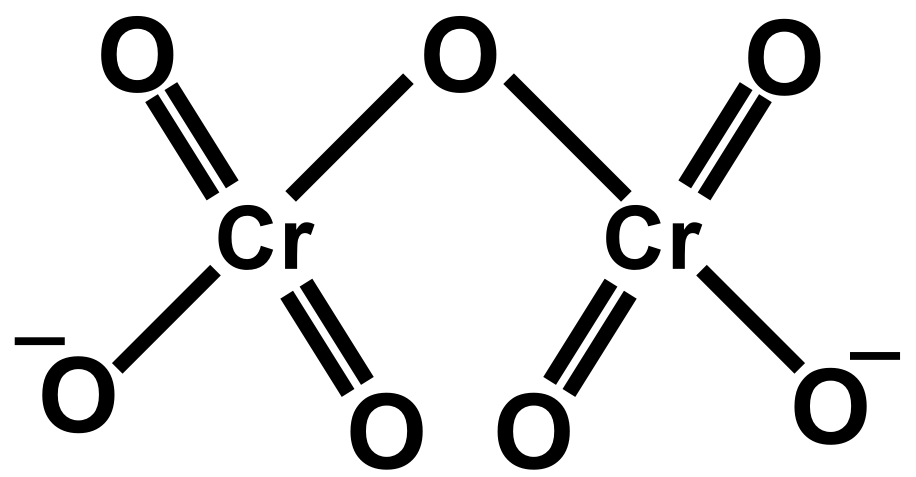
In dichromate dianion structure the negative charge can be on any oxygen attached to $Cr$ thus all the $Cr-O$ are equivalent and since one $Cr-O-Cr$ bond. Dichromate is a divalent inorganic anion obtained by removal of both protons from dichromic acid. It is a chromium oxoanion and a divalent inorganic anion. It is a conjugate base of a hydrogen dichromate.
Therefore, the correct answer is option D.
Note:
Remember that the resonance is a way of describing delocalized electrons within certain molecules or polyatomic ions where the bonding cannot be expressed by a single Lewis formula. A molecule or ion with such delocalized electrons is represented by several resonance structures.
Complete answer:
As we know the concept of chemical bonding and structures of ions. Also, the bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule. Bond lengths are expressed in terms of Angstrom or picometer. The two $Cr-O$ bonds which share the single oxygen atom are longer than the six $Cr-O$ terminal bonds that are equivalent. The salt of the dichromate ion is used for oxidation and reduction reactions. Dichromate poses an orange colour. The dichromate dianion consists of two tetrahedra sharing oxygen at the common corner.

In dichromate dianion structure the negative charge can be on any oxygen attached to $Cr$ thus all the $Cr-O$ are equivalent and since one $Cr-O-Cr$ bond. Dichromate is a divalent inorganic anion obtained by removal of both protons from dichromic acid. It is a chromium oxoanion and a divalent inorganic anion. It is a conjugate base of a hydrogen dichromate.
Therefore, the correct answer is option D.
Note:
Remember that the resonance is a way of describing delocalized electrons within certain molecules or polyatomic ions where the bonding cannot be expressed by a single Lewis formula. A molecule or ion with such delocalized electrons is represented by several resonance structures.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




