The wrong statement about ${{\text{N}}_2}{\text{O}}$ is that:
A. It is nitrous oxide.
B. It is least reactive oxide of nitrogen
C. It is not a linear molecule
D. It is known as laughing gas
Answer
591.9k+ views
Hint:${{\text{N}}_2}{\text{O}}$ is known as dinitrogen oxide. The oxidation state of nitrogen is $ + 1$. It is a stable oxide. Dinitrogen oxide shows $sp$ hybridization. It has an exhilarating effect on inhalation.
Complete answer:
Nitrogen forms various oxides such as nitrogen oxide, nitrogen dioxide, dinitrogen oxide, and dinitrogen trioxide and dinitrogen pentoxide.
The oxidation state of nitrogen in dinitrogen oxide is as follows:
The oxidation state of an oxygen atom is $ - 2$.
\[\left( {x\, \times 2} \right) + \left( { - 2\, \times 1} \right)\]
\[x = + 1\]
So, the oxidation state of nitrogen in dinitrogen oxide is \[ + 1\].
The nitrogen in \[ + 1\] oxidation state is known as nitrous. So, it is correct that dinitrogen oxide is nitrous oxide.
Octet of each atom of nitrous oxide is complete, so it is stable. Stability is indirectly proportional to the reactivity. Thus it is true that nitrous oxide is the least reactive oxide of nitrogen.
The structure of the nitrous oxide is as follows:
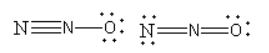
Nitrous oxide is a linear molecule, so the statement that it is not a linear molecule is not true.
The nitrous oxide on inhalation causes an intoxicating effect, so it is true that it is known as laughing gas.
Therefore option (C) It is not a linear molecule, is correct.
Note:
Nitrous oxide is used as anaesthesia in surgery. It has an analgesic effect. The hybridization of the nitrogen atom in nitrous oxide is sp. Nitrous oxide is diamagnetic. It is a colourless gas. It is neutral to a litmus test.
Complete answer:
Nitrogen forms various oxides such as nitrogen oxide, nitrogen dioxide, dinitrogen oxide, and dinitrogen trioxide and dinitrogen pentoxide.
The oxidation state of nitrogen in dinitrogen oxide is as follows:
The oxidation state of an oxygen atom is $ - 2$.
\[\left( {x\, \times 2} \right) + \left( { - 2\, \times 1} \right)\]
\[x = + 1\]
So, the oxidation state of nitrogen in dinitrogen oxide is \[ + 1\].
The nitrogen in \[ + 1\] oxidation state is known as nitrous. So, it is correct that dinitrogen oxide is nitrous oxide.
Octet of each atom of nitrous oxide is complete, so it is stable. Stability is indirectly proportional to the reactivity. Thus it is true that nitrous oxide is the least reactive oxide of nitrogen.
The structure of the nitrous oxide is as follows:
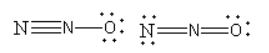
Nitrous oxide is a linear molecule, so the statement that it is not a linear molecule is not true.
The nitrous oxide on inhalation causes an intoxicating effect, so it is true that it is known as laughing gas.
Therefore option (C) It is not a linear molecule, is correct.
Note:
Nitrous oxide is used as anaesthesia in surgery. It has an analgesic effect. The hybridization of the nitrogen atom in nitrous oxide is sp. Nitrous oxide is diamagnetic. It is a colourless gas. It is neutral to a litmus test.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




