The sulphur molecule \[{{S}_{8}}\] can be represented as:
a.) Cubical structure
b.) Spherical structure
c.) Tetrahedral structure
d.) W shaped ring structure
Answer
618.3k+ views
Hint: Eight sulphur atoms form a puckered ring or crown structure to form an eight-atom molecule. Sulphur has six electrons in its outermost shell and requires two electrons to complete its octet state.
Complete step by step answer:
Sulphur has six electrons in its outermost shell and requires two electrons to complete its octet state. So, each sulphur atom shares two electrons, 1 with each adjoining sulphur atom
The atomic number (Z) of sulphur is sixteen and its electronic configuration is 2, 8, 6. The sulphur atom has six valence electrons. The chemical formula of sulphur molecules is \[{{S}_{8}}\] each sulphur atom is linked to similar atoms on either side by single covalent bonds and thus, completes its octet. The molecule is in the form of a ring also represented by crown shape.
So, the correct answer is “Option D”.
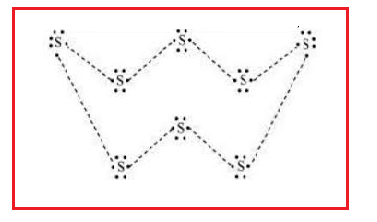
Note: Sulphur is abundant, multivalent, and non-metallic. Under normal conditions, Sulphur atoms form cyclic octatomic molecules with a chemical formula \[{{S}_{8}}\]. Elemental sulphur is a bright yellow crystalline solid at room temperature.
The most commonly encountered form of sulphur is the orthorhombic polymorph of \[{{S}_{8}}\], which adopts a puckered ring – or "crown" – structure. Two other polymorphs are known, also with nearly identical molecular structures. In addition to \[{{S}_{8}}\], sulphur rings of 6, 7, 9–15, 18 and 20 atoms are known. At least five allotropes are uniquely formed at high pressures, two of which are metallic. It is used in the manufacture of sulphuric acid, which in turn goes into fertilizers, batteries and cleaners. It's also used to refine oil and in processing ores. Pure sulphur has no smell.
Complete step by step answer:
Sulphur has six electrons in its outermost shell and requires two electrons to complete its octet state. So, each sulphur atom shares two electrons, 1 with each adjoining sulphur atom
The atomic number (Z) of sulphur is sixteen and its electronic configuration is 2, 8, 6. The sulphur atom has six valence electrons. The chemical formula of sulphur molecules is \[{{S}_{8}}\] each sulphur atom is linked to similar atoms on either side by single covalent bonds and thus, completes its octet. The molecule is in the form of a ring also represented by crown shape.
So, the correct answer is “Option D”.

Note: Sulphur is abundant, multivalent, and non-metallic. Under normal conditions, Sulphur atoms form cyclic octatomic molecules with a chemical formula \[{{S}_{8}}\]. Elemental sulphur is a bright yellow crystalline solid at room temperature.
The most commonly encountered form of sulphur is the orthorhombic polymorph of \[{{S}_{8}}\], which adopts a puckered ring – or "crown" – structure. Two other polymorphs are known, also with nearly identical molecular structures. In addition to \[{{S}_{8}}\], sulphur rings of 6, 7, 9–15, 18 and 20 atoms are known. At least five allotropes are uniquely formed at high pressures, two of which are metallic. It is used in the manufacture of sulphuric acid, which in turn goes into fertilizers, batteries and cleaners. It's also used to refine oil and in processing ores. Pure sulphur has no smell.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

Why is it 530 pm in india when it is 1200 afternoon class 10 social science CBSE

What is the full form of CNG A Complete Natural Gas class 10 social science CBSE

In cricket, what is a "Yorker" designed to do?

What is the full form of POSCO class 10 social science CBSE

Define Potential, Developed, Stock and Reserved resources




