The shape of sulphate ion is:
A. Hexagonal
B. Square planar
C. Trigonal bipyramidal
D. Tetrahedral
Answer
590.4k+ views
Hint: The sulphate ion is the anion with negative charge. The empirical formula of sulphate ion is $SO_4^{2 - }$ where the central atom is sulphur and it is bonded to two oxygen atoms with double bonds and two oxygen atoms with single bonds. The oxidation state of sulphate ion is -2.
Complete step by step answer:
The sulphate or the sulphate ion is a polyatomic anion which contains different atoms in the anion. The anions are the negatively charged species formed by the loss of electrons. The sulphate ion contains one sulphur atom and four oxygen atoms with -2 oxidation state. The empirical formula of the sulphate ion is $SO_4^{2 - }$. The sulphates are referred to as the salts of sulphuric acid and are prepared from sulphuric acid.
The shape of sulphate ion is shown below.
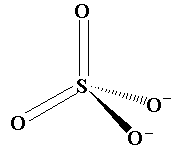
In this structure, the sulphur atom is bonded with two oxygen atoms with double bond and with two oxygen with single bond with -2 oxidation state.
The shape of the sulphate anion is tetrahedral as four bonds are present around the central atom and no lone pair is present on the central atom. The bond angle between the atoms bonded to the central atom is the 109.5 degree.
Thus, the shape of sulphate ions is tetrahedral.
Therefore, the correct option is C.
Note:
The VSEPR theory is applied to find out the geometry and the shape of the molecule depending on the bond pairs and lone pairs present in the structure. The shape of the chemical compound is also based on the hybrid orbital. The molecule or ion with tetrahedral shape has $s{p^3}$ hybridization at the central atom.
Complete step by step answer:
The sulphate or the sulphate ion is a polyatomic anion which contains different atoms in the anion. The anions are the negatively charged species formed by the loss of electrons. The sulphate ion contains one sulphur atom and four oxygen atoms with -2 oxidation state. The empirical formula of the sulphate ion is $SO_4^{2 - }$. The sulphates are referred to as the salts of sulphuric acid and are prepared from sulphuric acid.
The shape of sulphate ion is shown below.

In this structure, the sulphur atom is bonded with two oxygen atoms with double bond and with two oxygen with single bond with -2 oxidation state.
The shape of the sulphate anion is tetrahedral as four bonds are present around the central atom and no lone pair is present on the central atom. The bond angle between the atoms bonded to the central atom is the 109.5 degree.
Thus, the shape of sulphate ions is tetrahedral.
Therefore, the correct option is C.
Note:
The VSEPR theory is applied to find out the geometry and the shape of the molecule depending on the bond pairs and lone pairs present in the structure. The shape of the chemical compound is also based on the hybrid orbital. The molecule or ion with tetrahedral shape has $s{p^3}$ hybridization at the central atom.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




