The major product of the following is:
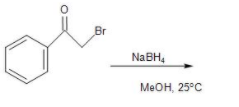
(A)

(B)
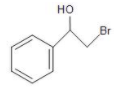
(C)
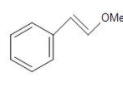
(D)





Answer
597.3k+ views
Hint: The carbonyl group present in the given compound, in presence of the selective reducing agent undergoes reduction with a formation of new C-H bond, followed by the protonation of the oxygen on the carbonyl carbon.
Complete step by step answer:
In the given compound, that is, 2-Bromoacetophenone the reaction taking place in presence of a reducing agent, that is sodium borohydride in methanol solvent. It causes the reduction of the ketone group present in the compound. In the mechanism, the hydride $({{H}^{-}})$ detaches from the sodium borohydride and attacks the partially positive carbonyl carbon of the ketone group. Thus, a new C-H bond is formed, followed by a negative charge on the oxygen. Thus, forming an alkoxide ion.
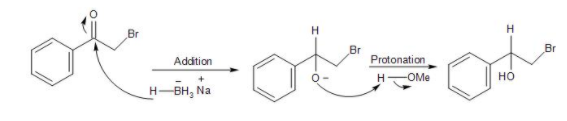
This is followed by the protonation of the ion by the methyl alcohol to form a secondary alcohol, that is, 2-Bromo-1-phenyl-ethanol compound. Thus, the reduction of the ketone to alcohol took place by the sodium borohydride in MeOH solvent.
Therefore, the major product formed from the reduction of the given compound is option (B) 2-Bromo-1-phenyl-ethanol.
Note: The $NaB{{H}_{4}}$ is a very selective reducing agent. It can only cause reduction of the aldehyde, ketones, and acid chloride to alcohols. Even though we get a primary alcohol in case of aldehydes and acid chlorides. Whereas the ketone generally forms secondary alcohol.
Also, due to the minimal difference in the electronegativity of boron (2.0) and hydrogen (2.1). The bond is less polar, thus less electron density of the hydrides. This makes them less nucleophilic and thus a mild reducing agent.
Complete step by step answer:
In the given compound, that is, 2-Bromoacetophenone the reaction taking place in presence of a reducing agent, that is sodium borohydride in methanol solvent. It causes the reduction of the ketone group present in the compound. In the mechanism, the hydride $({{H}^{-}})$ detaches from the sodium borohydride and attacks the partially positive carbonyl carbon of the ketone group. Thus, a new C-H bond is formed, followed by a negative charge on the oxygen. Thus, forming an alkoxide ion.

This is followed by the protonation of the ion by the methyl alcohol to form a secondary alcohol, that is, 2-Bromo-1-phenyl-ethanol compound. Thus, the reduction of the ketone to alcohol took place by the sodium borohydride in MeOH solvent.
Therefore, the major product formed from the reduction of the given compound is option (B) 2-Bromo-1-phenyl-ethanol.
Note: The $NaB{{H}_{4}}$ is a very selective reducing agent. It can only cause reduction of the aldehyde, ketones, and acid chloride to alcohols. Even though we get a primary alcohol in case of aldehydes and acid chlorides. Whereas the ketone generally forms secondary alcohol.
Also, due to the minimal difference in the electronegativity of boron (2.0) and hydrogen (2.1). The bond is less polar, thus less electron density of the hydrides. This makes them less nucleophilic and thus a mild reducing agent.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE

MABP stands for A Man and Biology Protection B Man class 12 biology CBSE

The correct structure of ethylenediaminetetraacetic class 12 chemistry CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Hormone responsible for the secretion of milk after class 12 biology CBSE




