The hybridization and geometry of $BrF3$ molecules are
A. $s{{p}^{3}}d$ and T shaped.
B. $s{{p}^{2}}{{d}^{2}}$ and tetragonal
C. $s{{p}^{3}}d$and bent
D. None of the above
Answer
598.2k+ views
Hint: Many theories have been formulated to know the nature of bonds in compounds like Valence bond theory, Molecular orbital theory, Crystal field theory, Valence shell electron pair repulsion theory.
The hybridization and geometry of $BrF3$ molecule is determined by VSEPR theory;i.e; valence shell electron pair repulsion theory.
Complete step by step solution:
-VSEPR theory states that the electron pairs repel each other, whether they are in the form of lone pair or bond pair. This is done in order to minimise the repulsion, thus increasing the stability of the compound.
-The knowledge of bond pair and lone pair of electrons gives us the idea of the geometry of the molecule. The geometry is found by counting the number of electron pairs and not the lone pairs.
-According to the electron pairs, the geometry can be decided as
-Molecular geometry, on the other hand, also depends on the number of the lone pairs. When there are no lone pairs, the denotation is simply $A{{X}_{n}}$ . When lone pairs are added, the denotation becomes $A{{X}_{n}}{{E}_{m}}$
where n=no. of bond pairs
m=no. of lone pairs
-The lone pairs are placed apart from each other while the bond pairs are placed apart from each other to ensure maximum stability. So the lone pairs are always placed on the equatorial plane first.
-$BrF3$ molecule contains 3 bonded electron pairs as one bromine atom is bonded with 3 fluorine atoms.
-If we observe the Bromine atom, it has a total 7 electrons in its outermost shell, three of which are bonded with flourine atoms. 4 electrons remain non-bonded and thus become the 2 lone pairs of electrons. These lone pairs have to be placed on the equatorial plane only.
-F atom needs only 1 more electron to reach the configuration of Ne which it does by bonding with Br. So, it completes its octet.
So now, Br has 3 bonded electron pairs and 2 lone pairs of electrons.
-The two lone pairs occupy the equatorial plane and 1 bond pair also gets that position as there can be 3 locations on the equatorial plane and 2 on the axial plane. The remaining 2 bond pairs are located on the axial plane. Thus the shape becomes T-shaped.
-Since there are total 5 electron pairs, the hybridization is $s{{p}^{3}}d$
The structure is shown as:
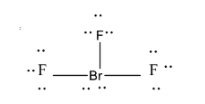
Therefore the correct option is A.
Note: Remember that there are 3 Br-F bonds which are covalent in nature as it involves sharing of electrons. Lone pairs of electrons are 2. Also, Br is the central atom and uses d orbital for the hybridization and this is the reason why the hybridization is $s{{p}^{3}}d$ and not $ds{{p}^{3}}$ .
The hybridization and geometry of $BrF3$ molecule is determined by VSEPR theory;i.e; valence shell electron pair repulsion theory.
Complete step by step solution:
-VSEPR theory states that the electron pairs repel each other, whether they are in the form of lone pair or bond pair. This is done in order to minimise the repulsion, thus increasing the stability of the compound.
-The knowledge of bond pair and lone pair of electrons gives us the idea of the geometry of the molecule. The geometry is found by counting the number of electron pairs and not the lone pairs.
-According to the electron pairs, the geometry can be decided as
| Number of electron pairs | Geometry of molecule |
| 2 | Linear |
| 3 | Trigonal planar |
| 4 | Tetrahedral |
| 5 | Trigonal bipyramidal |
| 6 | Octahedral |
-Molecular geometry, on the other hand, also depends on the number of the lone pairs. When there are no lone pairs, the denotation is simply $A{{X}_{n}}$ . When lone pairs are added, the denotation becomes $A{{X}_{n}}{{E}_{m}}$
where n=no. of bond pairs
m=no. of lone pairs
-The lone pairs are placed apart from each other while the bond pairs are placed apart from each other to ensure maximum stability. So the lone pairs are always placed on the equatorial plane first.
-$BrF3$ molecule contains 3 bonded electron pairs as one bromine atom is bonded with 3 fluorine atoms.
-If we observe the Bromine atom, it has a total 7 electrons in its outermost shell, three of which are bonded with flourine atoms. 4 electrons remain non-bonded and thus become the 2 lone pairs of electrons. These lone pairs have to be placed on the equatorial plane only.
-F atom needs only 1 more electron to reach the configuration of Ne which it does by bonding with Br. So, it completes its octet.
So now, Br has 3 bonded electron pairs and 2 lone pairs of electrons.
-The two lone pairs occupy the equatorial plane and 1 bond pair also gets that position as there can be 3 locations on the equatorial plane and 2 on the axial plane. The remaining 2 bond pairs are located on the axial plane. Thus the shape becomes T-shaped.
-Since there are total 5 electron pairs, the hybridization is $s{{p}^{3}}d$
The structure is shown as:

Therefore the correct option is A.
Note: Remember that there are 3 Br-F bonds which are covalent in nature as it involves sharing of electrons. Lone pairs of electrons are 2. Also, Br is the central atom and uses d orbital for the hybridization and this is the reason why the hybridization is $s{{p}^{3}}d$ and not $ds{{p}^{3}}$ .
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE




