The correct statement(s) regarding the following are:
(i) $HClO$ (ii) $HCl{{O}_{2}}$ (iii) $HCl{{O}_{3}}$ (iv) $HCl{{O}_{4}}$
(A) the number of $Cl=O$ bonds in (ii) and (iii) together is three
(B) the number of lone pairs of electrons on Cl in (ii) and (iii) together is three
(C) the hybridization of \[ClO\] in (iv) is $s{{p}^{3}}$
(D) amongst (i) to (iv), the strongest acid is (i)
Answer
618k+ views
Hint: Draw the structure of the molecules. Determine the number of double bonds present in them. Also, determine the number of lone pairs on chlorine atoms in each structure. Find out the oxidation states of Cl atoms in each structure.
Complete answer:
Chlorine forms four types of oxyacids:
-Hypohalous acid: Hypochlorous acid

Hypochlorous acid has one oxygen atom containing two lone pairs of electrons and one chlorine atom containing three lone pairs of electrons. There are no double bonds present in the molecule.
\[\text{Oxidation state }=\text{ }+1\]
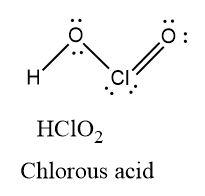
Chlorous acid has two oxygen atoms containing two lone pairs of electrons each and one chlorine atom containing two lone pairs of electrons. There is one $Cl=O$ double bond present in the molecule.
\[\text{Oxidation state }=\text{ }+3\]
-Halic acid: Chloric acid
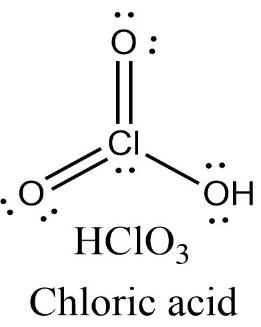
Chloric acid has three oxygen atoms containing two lone pairs of electrons each and one chlorine atom containing one lone pair of electrons. There are two $Cl=O$ double bonds present in the molecule.
\[\text{Oxidation state }=\text{ }+3\]
-Perhalic acid: Perchloric acid
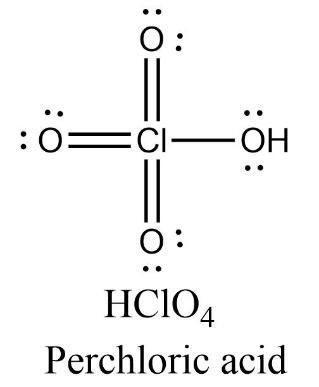
Perchloric acid has four oxygen atoms containing two lone pairs of electrons each and one chlorine atom containing no lone pair of electrons. There are three $Cl=O$ double bonds present in the molecule.
\[\text{Oxidation state }=\text{ }+5\]
Now, let’s take a look at the options given in the question.
(A) It is correct because the number of $Cl=O$ bonds in $HCl{{O}_{2}}$ and $HCl{{O}_{3}}$ together is three.
(B) It is correct because the number of lone pairs of electrons in $HCl{{O}_{2}}$ and $HCl{{O}_{3}}$ together is three.
(C) It is incorrect because the hybridization of $Cl=O$ in $HCl{{O}_{4}}$ is $s{{p}^{2}}$.
(D) We know that as the oxidation state increases, acidity increases. In this case, the oxidation state of Perchloric acid is +7. So, Perchloric acid is the strongest acid amongst other oxyacids of chlorine.
Order of Acidity can be given as,
$HCl{{O}_{4}} > HCl{{O}_{3}} > HCl{{O}_{2}} > HClO$
So, we can conclude that option (D) is also incorrect because it states hypochlorous acid is the strongest acid.
So, the correct answer is “Option A and B”.
Note: Mistakes can happen while drawing the structure of molecules. Remember, a doubly bonded oxygen atom has two lone pairs and a singly bonded chlorine atom has three lone pairs.
Complete answer:
Chlorine forms four types of oxyacids:
-Hypohalous acid: Hypochlorous acid

Hypochlorous acid has one oxygen atom containing two lone pairs of electrons and one chlorine atom containing three lone pairs of electrons. There are no double bonds present in the molecule.
\[\text{Oxidation state }=\text{ }+1\]

Chlorous acid has two oxygen atoms containing two lone pairs of electrons each and one chlorine atom containing two lone pairs of electrons. There is one $Cl=O$ double bond present in the molecule.
\[\text{Oxidation state }=\text{ }+3\]
-Halic acid: Chloric acid

Chloric acid has three oxygen atoms containing two lone pairs of electrons each and one chlorine atom containing one lone pair of electrons. There are two $Cl=O$ double bonds present in the molecule.
\[\text{Oxidation state }=\text{ }+3\]
-Perhalic acid: Perchloric acid

Perchloric acid has four oxygen atoms containing two lone pairs of electrons each and one chlorine atom containing no lone pair of electrons. There are three $Cl=O$ double bonds present in the molecule.
\[\text{Oxidation state }=\text{ }+5\]
Now, let’s take a look at the options given in the question.
(A) It is correct because the number of $Cl=O$ bonds in $HCl{{O}_{2}}$ and $HCl{{O}_{3}}$ together is three.
(B) It is correct because the number of lone pairs of electrons in $HCl{{O}_{2}}$ and $HCl{{O}_{3}}$ together is three.
(C) It is incorrect because the hybridization of $Cl=O$ in $HCl{{O}_{4}}$ is $s{{p}^{2}}$.
(D) We know that as the oxidation state increases, acidity increases. In this case, the oxidation state of Perchloric acid is +7. So, Perchloric acid is the strongest acid amongst other oxyacids of chlorine.
Order of Acidity can be given as,
$HCl{{O}_{4}} > HCl{{O}_{3}} > HCl{{O}_{2}} > HClO$
So, we can conclude that option (D) is also incorrect because it states hypochlorous acid is the strongest acid.
So, the correct answer is “Option A and B”.
Note: Mistakes can happen while drawing the structure of molecules. Remember, a doubly bonded oxygen atom has two lone pairs and a singly bonded chlorine atom has three lone pairs.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Which gas is abundant in air class 11 chemistry CBSE

10 examples of friction in our daily life




