The correct order of decreasing acidity of nitrophenols will be:
a. m-nitrophenol
 p-nitrophenol
p-nitrophenol
 o-nitrophenol
o-nitrophenol
b. o-nitrophenol
 m-nitrophenol
m-nitrophenol
 p-nitrophenol
p-nitrophenol
c. p-nitrophenol
 m-nitrophenol
m-nitrophenol
 o-nitrophenol
o-nitrophenol
d. p-nitrophenol
 o-nitrophenol
o-nitrophenol
 m-nitrophenol
m-nitrophenol








Answer
551.5k+ views
Hint: The general formula of nitrophenols is: ${ HOC }_{ 6 }{ H }_{ 5-x }{ (NO }_{ 2 }{ ) }_{ x }$
The structure of nitrophenol is shown below:
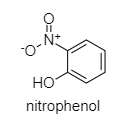
Here the acidity depends on the presence of electron-withdrawing or electron releasing groups and their respective positions.
Complete step-by-step answer:
An inductive effect is an electronic effect due to the polarisation of σ bonds within a molecule or ion. This is typically due to an electronegativity difference between the atoms at either end of the bond.
${ -NO }_{ 2 }$ is an electron-withdrawing group.
${ -NO }_{ 2 }$ group at ortho and para position pulls back electrons of the OH bond towards itself by the stronger -R effect while the ${ -NO }_{ 2 }$ group at m-position pulls back electrons of the OH bond by the weaker effect.
Thus, ortho and para-nitrophenols are more acidic than m-nitrophenol is a little less acidic than p-nitrophenol because of intramolecular hydrogen bonding which makes the loss of a proton difficult to remove.
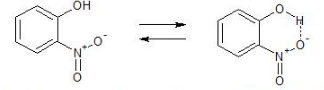
Figure: Showing intramolecular hydrogen bonding in m-nitrophenol.
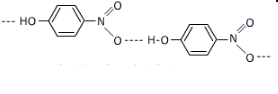
Figure: Showing intermolecular hydrogen bonding in p-nitrophenol
Hence, The correct order of decreasing acidity of nitrophenols will be:
p-nitrophenol
 o-nitrophenol
o-nitrophenol
 m-nitrophenol
m-nitrophenol
The correct option is D.
Additional Information:
Resonance effect: It can be defined as the delocalization of π electrons due to the presence of electron releasing or electron-withdrawing substituents, which results in canonical forms with different electron distribution.
It is designated as the R or M effect.
Application: It gives extra stability to the compound by delocalization of charges. Some time it activates or deactivates the benzene ring towards electrophilic substitution.
Note: The possibility to make a mistake is that you may choose option C. But p-nitrophenol shows both -R and -I effects, so it will be more acidic than m-nitrophenol as it does not show any such effect.
The structure of nitrophenol is shown below:

Here the acidity depends on the presence of electron-withdrawing or electron releasing groups and their respective positions.
Complete step-by-step answer:
An inductive effect is an electronic effect due to the polarisation of σ bonds within a molecule or ion. This is typically due to an electronegativity difference between the atoms at either end of the bond.
${ -NO }_{ 2 }$ is an electron-withdrawing group.
${ -NO }_{ 2 }$ group at ortho and para position pulls back electrons of the OH bond towards itself by the stronger -R effect while the ${ -NO }_{ 2 }$ group at m-position pulls back electrons of the OH bond by the weaker effect.
Thus, ortho and para-nitrophenols are more acidic than m-nitrophenol is a little less acidic than p-nitrophenol because of intramolecular hydrogen bonding which makes the loss of a proton difficult to remove.

Figure: Showing intramolecular hydrogen bonding in m-nitrophenol.

Figure: Showing intermolecular hydrogen bonding in p-nitrophenol
Hence, The correct order of decreasing acidity of nitrophenols will be:
p-nitrophenol


The correct option is D.
Additional Information:
Resonance effect: It can be defined as the delocalization of π electrons due to the presence of electron releasing or electron-withdrawing substituents, which results in canonical forms with different electron distribution.
It is designated as the R or M effect.
Application: It gives extra stability to the compound by delocalization of charges. Some time it activates or deactivates the benzene ring towards electrophilic substitution.
Note: The possibility to make a mistake is that you may choose option C. But p-nitrophenol shows both -R and -I effects, so it will be more acidic than m-nitrophenol as it does not show any such effect.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




