Sulphur is used in vulcanization of rubbers.
a. True
b. False
Answer
581.4k+ views
Hint: Vulcanization is the term used for the process of hardening natural rubber and stiffening it, in order to improve its properties and make it more durable. This mainly happens by the crosslinking of its monomer units which increase hardness.
Colloidal particles possess charge in them.
Complete step by step answer:
Vulcanization of rubber is the process of heating rubber in presence of Sulphur. The crude (raw) rubber is heated with 3 percent Sulphur at about 1200 degree Celsius for 2-3 hours. Natural rubber is not very strong in nature and therefore, it softens on heating. An additive can be added at this step.
- As a result of this, the chains of rubber and sulphur cross-link at reactive sites of double bonds with each other to form a strong and stable polymer.
The reaction taking place is as shown below,
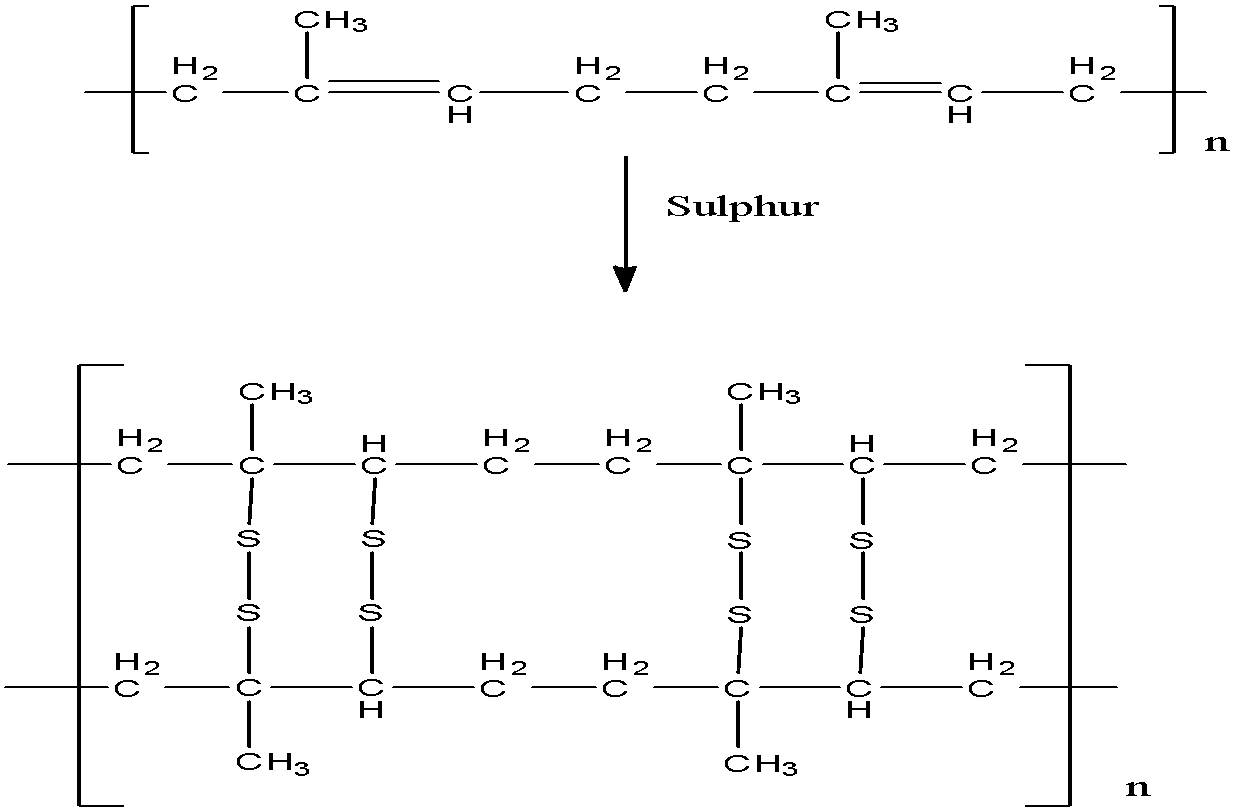
The temperature should be taken care of. At higher temperature (above 335 K) rubber becomes soft, whereas at lower temperature, rubber becomes brittle and it also shows high water absorption capacity. The correct option is option “A” .
Additional Information :Natural rubber is also known as cis-1,4-polyisoprene as it is made by polymerization of isoprene.
Note: Rubbers are categorized as natural and synthetic. Natural rubbers are the elastomers that are obtained naturally. It is composed of isoprene monomers whereas synthetic rubbers are derived from petroleum, natural gas and polymerization of butadiene.
Colloidal particles possess charge in them.
Complete step by step answer:
Vulcanization of rubber is the process of heating rubber in presence of Sulphur. The crude (raw) rubber is heated with 3 percent Sulphur at about 1200 degree Celsius for 2-3 hours. Natural rubber is not very strong in nature and therefore, it softens on heating. An additive can be added at this step.
- As a result of this, the chains of rubber and sulphur cross-link at reactive sites of double bonds with each other to form a strong and stable polymer.
The reaction taking place is as shown below,

The temperature should be taken care of. At higher temperature (above 335 K) rubber becomes soft, whereas at lower temperature, rubber becomes brittle and it also shows high water absorption capacity. The correct option is option “A” .
Additional Information :Natural rubber is also known as cis-1,4-polyisoprene as it is made by polymerization of isoprene.
Note: Rubbers are categorized as natural and synthetic. Natural rubbers are the elastomers that are obtained naturally. It is composed of isoprene monomers whereas synthetic rubbers are derived from petroleum, natural gas and polymerization of butadiene.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




