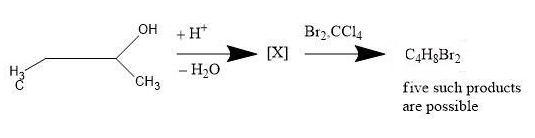
How many structures of X is possible?
(A) 4
(B) 5
(C) 6
(D) 3

Answer
597.3k+ views
Hint: The intermediate structures for any given reaction give some basic reaction rules that would be vital and sensible.
Some of these rules are resonance effect, inductive effect, etc.
Complete step by step solution:
The given reaction is about the dehydrogenation of Butan-2-ol. Let us see the mechanism and conditions for the reaction.
There are 2 steps for the given reaction i.e.
1. The positive ion removes a water molecule from the butan-2-ol and carbocation is formed.
2. The carbocation loses the hydrogen ion to form a double bond.
The structures arising from the second step will be the answer for the given problem.
When the carbocation loses the hydrogen ion (from adjacent carbon atom of the carbon atom already having positive charge) to form double bond, it has two choices i.e.
1. Lose the hydrogen ion from the adjacent $C{{H}_{3}}$ group.
-Due to this move, But-1-ene ($C{{H}_{3}}-C{{H}_{2}}-CH=C{{H}_{2}}$) is formed.
2. Loss the hydrogen ion from the adjacent $C{{H}_{2}}$ group.
-Due to this move, But-2-ene ($C{{H}_{3}}-CH=CH-C{{H}_{3}}$) is formed.
-But, here the effect of resonance will take place as But-2-ene has a geometrical isomer. We get a mixture of two isomers i.e. cis-but-2-ene and trans-but-2-ene.
Thus,
Dehydration of Butan-2-ol leads to mixture containing,
a. But-1-ene
b. cis-but-2-ene
c. trans-but-2-ene
Therefore, three types of intermediates or structures would be formed for the given dehydration of the But-2-ol.
Hence, option (D) is correct.
Note: Do note to consider the phenomenon of resonance when we see the possibilities of having geometrical isomers.The loss of hydrogen ion from a carbocation always takes place from the alpha carbon atom of carbon holding the positive charge. Thus, the number of alpha carbons decides the basic number of intermediate structures having double bonds within themselves.
Some of these rules are resonance effect, inductive effect, etc.
Complete step by step solution:
The given reaction is about the dehydrogenation of Butan-2-ol. Let us see the mechanism and conditions for the reaction.
There are 2 steps for the given reaction i.e.
1. The positive ion removes a water molecule from the butan-2-ol and carbocation is formed.
2. The carbocation loses the hydrogen ion to form a double bond.
The structures arising from the second step will be the answer for the given problem.
When the carbocation loses the hydrogen ion (from adjacent carbon atom of the carbon atom already having positive charge) to form double bond, it has two choices i.e.
1. Lose the hydrogen ion from the adjacent $C{{H}_{3}}$ group.
-Due to this move, But-1-ene ($C{{H}_{3}}-C{{H}_{2}}-CH=C{{H}_{2}}$) is formed.
2. Loss the hydrogen ion from the adjacent $C{{H}_{2}}$ group.
-Due to this move, But-2-ene ($C{{H}_{3}}-CH=CH-C{{H}_{3}}$) is formed.
-But, here the effect of resonance will take place as But-2-ene has a geometrical isomer. We get a mixture of two isomers i.e. cis-but-2-ene and trans-but-2-ene.
Thus,
Dehydration of Butan-2-ol leads to mixture containing,
a. But-1-ene
b. cis-but-2-ene
c. trans-but-2-ene
Therefore, three types of intermediates or structures would be formed for the given dehydration of the But-2-ol.
Hence, option (D) is correct.
Note: Do note to consider the phenomenon of resonance when we see the possibilities of having geometrical isomers.The loss of hydrogen ion from a carbocation always takes place from the alpha carbon atom of carbon holding the positive charge. Thus, the number of alpha carbons decides the basic number of intermediate structures having double bonds within themselves.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

What are the major means of transport Explain each class 12 social science CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




