Structure of $Xe{F_6}$ is octahedron with $s{p^3}{d^3}$ hybridization of $Xe$ .
A.True
B.False
Answer
588.6k+ views
Hint:We can find out the hybridization of an atom in a compound, utilizing a steric number. We can compute the steric number by summarizing the number of atoms attached to the center molecule and the solitary sets of electrons on the central metal particles. Tell us that on the off chance that the steric number is 4, at that point we state the molecule is in $s{p^3}$ hybridization, on the off chance that the steric number is 3, at that point, it is $s{p^2}$ hybridization, if the steric number is 2, at that point it is $sp$ hybridization.
Complete answer:
We can draw the structure of $Xe{F_6}$ as,
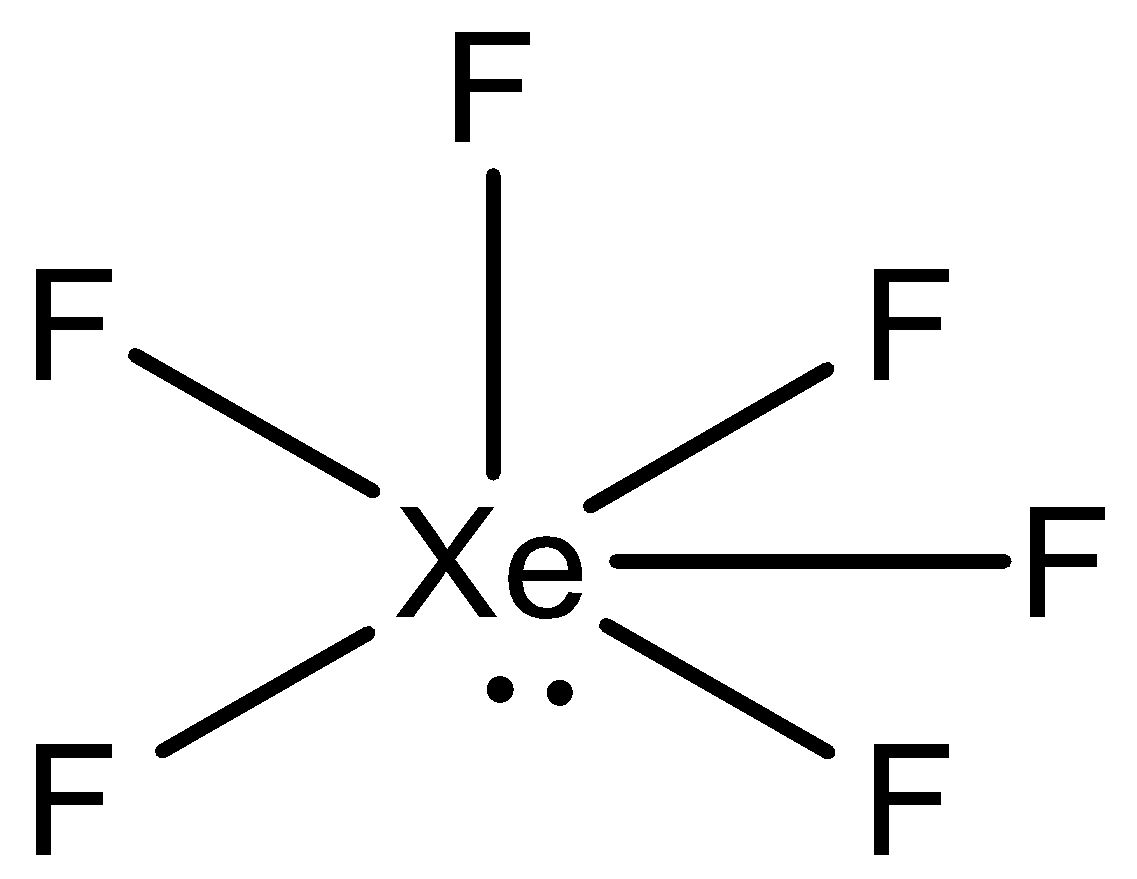
During the arrangement of $Xe{F_6}$ , xenon has eight electrons in its valence shell and it creates six bonds with the fluorine atoms. Further, the atoms will have one lone pair of electrons and six bond pairs. Presently on the off chance that we take the steric number, at that point, it will be seven. This can be deciphered as $s{p^3}{d^3}$ hybridization.
After hybridization, $Xe{F_6}$ molecular geometry would be distorted octahedral or square bipyramidal. What takes place here is that the atoms of fluorine atoms are located in the apexes of the octahedron while the lone pairs travel in the space to evade or decrease the repulsion.
Therefore, the option (B) is correct.
Note:
We have to remember that, $Xe{F_6}$ contains seven pairs of electrons. In that the number of bond pairs is six and the number of lone pairs is one. The number of electrons in the outermost shell of xenon is eight. When the fluorides of xenon have made electrons in the outermost shell of xenon get unpaired and are moved to empty $5d$ orbital.
Complete answer:
We can draw the structure of $Xe{F_6}$ as,

During the arrangement of $Xe{F_6}$ , xenon has eight electrons in its valence shell and it creates six bonds with the fluorine atoms. Further, the atoms will have one lone pair of electrons and six bond pairs. Presently on the off chance that we take the steric number, at that point, it will be seven. This can be deciphered as $s{p^3}{d^3}$ hybridization.
After hybridization, $Xe{F_6}$ molecular geometry would be distorted octahedral or square bipyramidal. What takes place here is that the atoms of fluorine atoms are located in the apexes of the octahedron while the lone pairs travel in the space to evade or decrease the repulsion.
Therefore, the option (B) is correct.
Note:
We have to remember that, $Xe{F_6}$ contains seven pairs of electrons. In that the number of bond pairs is six and the number of lone pairs is one. The number of electrons in the outermost shell of xenon is eight. When the fluorides of xenon have made electrons in the outermost shell of xenon get unpaired and are moved to empty $5d$ orbital.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




