State true or false – The conductivity of an intrinsic semiconductor decreases with increase in temperature.
A) True
B) False
Answer
597k+ views
Hint: The conductivity of a material depends upon the number of free electrons present in the conduction band of the material. For electrons to be present in the conduction band, they must overcome the energy band gap between the valence band and conduction band and jump up to the conduction band.
Complete step by step answer:
The conductivity of a substance is a measure of how easily it allows current to flow through it. The greater the conductivity of a substance, more easily will current flow through it.
The conductivity of a material depends upon the number of free electrons present in the conduction band of the substance as these electrons are responsible for current flow. For electrons to reach the conduction band they must overcome the energy band gap between the conduction and valence band.
For semiconductors, the band gap between the conduction band and the valence band is low. Therefore, when the temperature rises, the electrons in the valence band gain thermal energy and can jump up to the conduction band by overcoming the energy gap. Therefore, this leads to an increase in the number of free electrons in the conduction band and eventually, increase in the conductivity of the semiconductor.
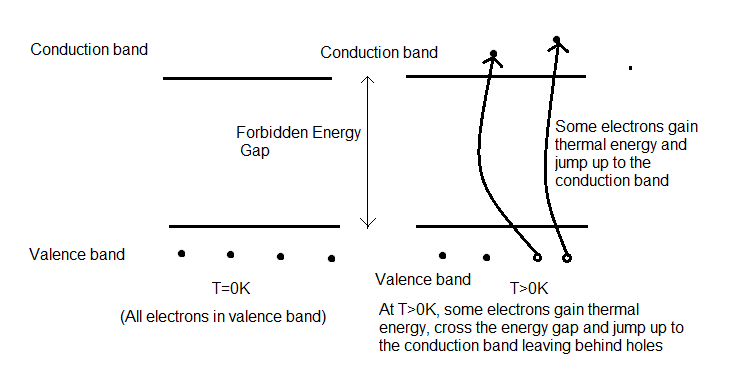
Therefore, the correct option is B) False.
Note:
The conductivity of semiconductors increases with rise in temperature but, for metals, it decreases with rise in temperature. This is because the energy gap between the conduction and valence band is zero as both the bands overlap. Therefore, no extra electrons can jump up to the conduction band by overcoming the energy gap since all of them are already present in it. However, the greater thermal energy leads to greater vibrations in the atoms of the metal, which hinder the flow of electrons and hence, hinder current flow. Therefore, the conductivity of metals decreases with temperature.
Students must be aware of this difference in the behavior of the conductivities of metals and semiconductors with increase in temperature.
Complete step by step answer:
The conductivity of a substance is a measure of how easily it allows current to flow through it. The greater the conductivity of a substance, more easily will current flow through it.
The conductivity of a material depends upon the number of free electrons present in the conduction band of the substance as these electrons are responsible for current flow. For electrons to reach the conduction band they must overcome the energy band gap between the conduction and valence band.
For semiconductors, the band gap between the conduction band and the valence band is low. Therefore, when the temperature rises, the electrons in the valence band gain thermal energy and can jump up to the conduction band by overcoming the energy gap. Therefore, this leads to an increase in the number of free electrons in the conduction band and eventually, increase in the conductivity of the semiconductor.

Therefore, the correct option is B) False.
Note:
The conductivity of semiconductors increases with rise in temperature but, for metals, it decreases with rise in temperature. This is because the energy gap between the conduction and valence band is zero as both the bands overlap. Therefore, no extra electrons can jump up to the conduction band by overcoming the energy gap since all of them are already present in it. However, the greater thermal energy leads to greater vibrations in the atoms of the metal, which hinder the flow of electrons and hence, hinder current flow. Therefore, the conductivity of metals decreases with temperature.
Students must be aware of this difference in the behavior of the conductivities of metals and semiconductors with increase in temperature.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




