Reduction of lactol S with sodium borohydride gives:
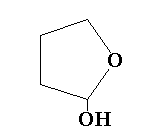
A.
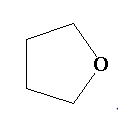
B.
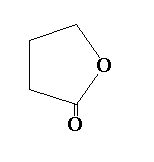
C.
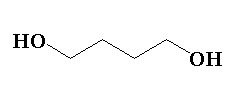
D.
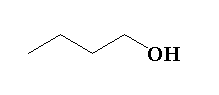





Answer
601.5k+ views
Hint: In this reaction lactol is converted to a compound containing hydroxyl group. The sodium borohydride is a reducing agent which is used to convert the carbonyl group (Aldehyde or ketone) into alcohol.
Complete step by step answer:
The lactol S is a five membered ring structure containing four carbon and an oxygen atom. The hydroxyl group is attached to the carbon adjacent to the oxygen atom.
The lactol is formed by the intramolecular nucleophilic addition of the hydroxyl group to the carbonyl group of an aldehydic compound or ketone containing compound. Here, the hydroxyl group acts as a nucleophile. In this reaction sodium borohydride is used which is a reducing agent. The sodium borohydride \[NaB{H_4}\], contains sodium as \[N{a^ + }\] and borohydride as \[BH_4^ -\]. The negative hydrogen will attack the carbonyl carbon and bond will shift forming a negative charge on the oxygen. Further the negative charge of the oxygen will abstract the hydrogen of the water and hydroxide ion will be released. The hydroxide ion will then act as a nucleophile and will attack the positive sodium ion to form water and the main product will be formed. The main product formed will be butane-1,4-diol.
The reaction of lactol S with sodium borohydride is shown below.
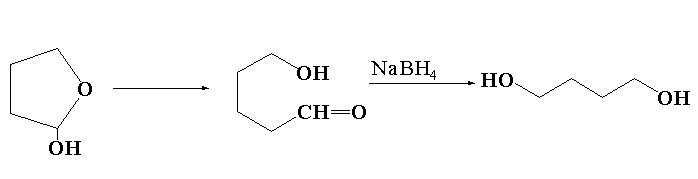
In this reaction, lactol S reacts with sodium borohydride to form butane-1,4-diol.
Therefore, the correct option is C.
Note:
The water used in this reaction acts as a solvent. The sodium borohydride is a reducing agent where the hydrogen anion will attack the carbonyl carbon. Don’t get confused as lactol contains a hydroxyl group.
Complete step by step answer:
The lactol S is a five membered ring structure containing four carbon and an oxygen atom. The hydroxyl group is attached to the carbon adjacent to the oxygen atom.
The lactol is formed by the intramolecular nucleophilic addition of the hydroxyl group to the carbonyl group of an aldehydic compound or ketone containing compound. Here, the hydroxyl group acts as a nucleophile. In this reaction sodium borohydride is used which is a reducing agent. The sodium borohydride \[NaB{H_4}\], contains sodium as \[N{a^ + }\] and borohydride as \[BH_4^ -\]. The negative hydrogen will attack the carbonyl carbon and bond will shift forming a negative charge on the oxygen. Further the negative charge of the oxygen will abstract the hydrogen of the water and hydroxide ion will be released. The hydroxide ion will then act as a nucleophile and will attack the positive sodium ion to form water and the main product will be formed. The main product formed will be butane-1,4-diol.
The reaction of lactol S with sodium borohydride is shown below.

In this reaction, lactol S reacts with sodium borohydride to form butane-1,4-diol.
Therefore, the correct option is C.
Note:
The water used in this reaction acts as a solvent. The sodium borohydride is a reducing agent where the hydrogen anion will attack the carbonyl carbon. Don’t get confused as lactol contains a hydroxyl group.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In order to find out the different types of gametes class 12 biology NEET_UG

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




