What is the radius of Sodium atom if it crystallizes in BCC unit cell edge length of $ 400pm $?
Answer
507.3k+ views
Hint: The unit cell is the smallest repeating unit of a crystal lattice. There are different types of crystal lattices, out of which one is body-centered cubic BCC which can be seen in the below image:
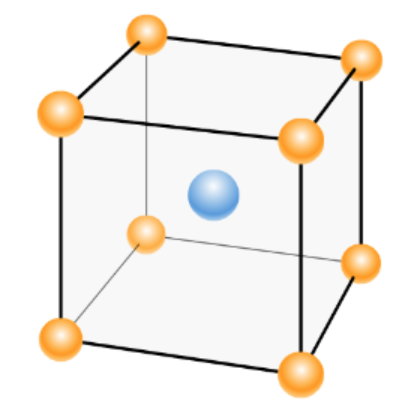
When any atoms are arranged in a body-centered cubic cell, the radius will be determined by substituting the edge length in the below formula.
$ r = \dfrac{{\sqrt 3 }}{4} \times a $
$ r $ is radius of a body centered cubic unit cell
$ a $ is edge length.
Complete answer:
Given that the sodium atom is crystallized in a body-centered cubic cell, with the edge length of $ 400pm $.
We know the radius formula in BCC as
$ r = \dfrac{{\sqrt 3 }}{4} \times a $
Substituting this edge length in the above formula,
$ r = \dfrac{{\sqrt 3 }}{4} \times 400 $
On simplification, we get the radius as
$ r = 173.2pm $
Thus, the radius of the sodium atom if it crystallizes in BCC unit cell edge length of $ 400pm $ is $ 173.2pm $ .
Additional information:
• Crystallography is a branch that deals mainly with crystal structures. According to this concept, the crystal structure is an order of the arrangement of atoms, ions, or molecules.
• The crystal lattice is also known as crystal structure which is nothing but the arrangement of atoms, ions, or molecules in the form of a space lattice.
• They are different types of crystal structures like body-centered cubic (BCC), face-centered cubic (FCC), and hexagonal cubic packing (HCP).
Note:
• While calculating the radius of a crystal lattice, the edge length is usually taken in the units of picometers as these measurements are very minute.
• 1 picometer is equal to $10^{-12}$ meters. Picometers can be simply represented as $ pm $.
• If the edge length is given in meters or angstroms, conversion should be made. Where one angstrom is equal to $ 100 $ picometers.

When any atoms are arranged in a body-centered cubic cell, the radius will be determined by substituting the edge length in the below formula.
$ r = \dfrac{{\sqrt 3 }}{4} \times a $
$ r $ is radius of a body centered cubic unit cell
$ a $ is edge length.
Complete answer:
Given that the sodium atom is crystallized in a body-centered cubic cell, with the edge length of $ 400pm $.
We know the radius formula in BCC as
$ r = \dfrac{{\sqrt 3 }}{4} \times a $
Substituting this edge length in the above formula,
$ r = \dfrac{{\sqrt 3 }}{4} \times 400 $
On simplification, we get the radius as
$ r = 173.2pm $
Thus, the radius of the sodium atom if it crystallizes in BCC unit cell edge length of $ 400pm $ is $ 173.2pm $ .
Additional information:
• Crystallography is a branch that deals mainly with crystal structures. According to this concept, the crystal structure is an order of the arrangement of atoms, ions, or molecules.
• The crystal lattice is also known as crystal structure which is nothing but the arrangement of atoms, ions, or molecules in the form of a space lattice.
• They are different types of crystal structures like body-centered cubic (BCC), face-centered cubic (FCC), and hexagonal cubic packing (HCP).
Note:
• While calculating the radius of a crystal lattice, the edge length is usually taken in the units of picometers as these measurements are very minute.
• 1 picometer is equal to $10^{-12}$ meters. Picometers can be simply represented as $ pm $.
• If the edge length is given in meters or angstroms, conversion should be made. Where one angstrom is equal to $ 100 $ picometers.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE




