P-V diagram of an ideal gas is given in the figure
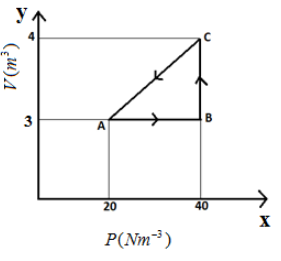
Work done on the gas in process CA is
A. $60J$
B. $70J$
C. $30J$
D. $20J$

Answer
600.9k+ views
Hint:In a cyclic process, the total work done is equal to the area enclosed by the graph. The work done will be positive if the cycle is going in clockwise direction and negative if the cycle is going in an anti-clockwise direction.
Formula used:
$W = \dfrac{{({P_f} + {P_i})}}{2}\Delta V$
Where $W$ is the total work done, ${P_i}$ is the initial pressure, ${P_f}$ is the final pressure and $\Delta V$ is the change in volume.
Complete step by step solution:
In the above cyclic process, while going from C to A, we can notice that it is a straight line that is both the volume and pressure are changing. We can obtain the total work done in this in a similar way we find the work done for an adiabatic process which is $P\Delta V$ where $P$ is the pressure and $\Delta V$ is the change in volume of the gas. But instead of constant pressure $P$, we consider the average pressure $\dfrac{{({P_f} + {P_i})}}{2}$ .
Thus we obtain the formula $W = \dfrac{{({P_f} + {P_i})}}{2}\Delta V$ .
It the above figure, it is given the value of initial pressure ${P_i}$ as $20N{m^{ - 3}}$ , final pressure ${P_f}$ as $40N{m^{ - 3}}$ and we can obtain the change in volume $\Delta V$ as $1{m^3}$ .
By substituting these values in the above formula, we get
$W = \left( {\dfrac{{20 + 40}}{2}} \right) \times 1 = 30J$
So,the correct option is C.
Note:For a cyclic process that is going in anti-clockwise direction, the total work is negative. The total work done can be found by summing up all the works done in A to B, B to C and C to A or by finding the area enclosed by the graph.
Formula used:
$W = \dfrac{{({P_f} + {P_i})}}{2}\Delta V$
Where $W$ is the total work done, ${P_i}$ is the initial pressure, ${P_f}$ is the final pressure and $\Delta V$ is the change in volume.
Complete step by step solution:
In the above cyclic process, while going from C to A, we can notice that it is a straight line that is both the volume and pressure are changing. We can obtain the total work done in this in a similar way we find the work done for an adiabatic process which is $P\Delta V$ where $P$ is the pressure and $\Delta V$ is the change in volume of the gas. But instead of constant pressure $P$, we consider the average pressure $\dfrac{{({P_f} + {P_i})}}{2}$ .
Thus we obtain the formula $W = \dfrac{{({P_f} + {P_i})}}{2}\Delta V$ .
It the above figure, it is given the value of initial pressure ${P_i}$ as $20N{m^{ - 3}}$ , final pressure ${P_f}$ as $40N{m^{ - 3}}$ and we can obtain the change in volume $\Delta V$ as $1{m^3}$ .
By substituting these values in the above formula, we get
$W = \left( {\dfrac{{20 + 40}}{2}} \right) \times 1 = 30J$
So,the correct option is C.
Note:For a cyclic process that is going in anti-clockwise direction, the total work is negative. The total work done can be found by summing up all the works done in A to B, B to C and C to A or by finding the area enclosed by the graph.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

How many of the following diseases can be studied with class 11 biology CBSE

Which of the following enzymes is used for carboxylation class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE




