What is the proper name of the compound above?
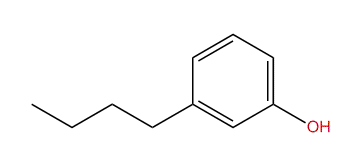
A.1-phenylethane
B.m-butyl hydroxy benzene
C.o-methyl hydroxybenzene
D.m-butylphenol

Answer
540.3k+ views
Hint: The growing number of organic compounds found each day, along with the fact that many of these compounds are isomers of other compounds, necessitates the creation of a formal nomenclature scheme. Each distinct compound must be assigned a distinctive and unique name, just as each distinct compound has a unique molecular structure that can be designated by a structural formula.
Complete answer:
Substituted benzene ring compounds have a less systematic nomenclature than alkanes, alkenes, and alkynes. As seen by the combined names mentioned below, a few mono-substituted compounds are named by using a group name as a prefix to "benzene." The rest of these molecules, on the other hand, have one-of-a-kind names. In order to learn these titles, memorization is the only option.
Phenyl, abbreviated Ph-, and benzyl, abbreviated Bn-, are two frequently found substituent groups that incorporate a benzene ring. These are shown along with illustrations of how they should be used. A phenyl group should not be confused with the compound phenol. Ar- for an aryl group is a general and useful standardised notation that complements the use of R- for an alkyl group (any aromatic ring).
Where there are several substituents on a benzene ring, the relative positions of the substituents must be shown by counting the ring carbons or by some other method. The prefixes ortho, meta, and para are generally used to denote a 1,2-, 1,3-, or 1,4- relationship in the case of disubstituted benzenes. The first row of compounds in the following illustrations shows this use in colour. Any disubstituted toluenes have singular names (for example, xylene, cresol, and toluidine), and their isomers are usually labelled with the ortho, meta, or para prefix. Relevant isomers of a few disubstituted benzenes have singular names (e.g. salicylic acid & resorcinol).
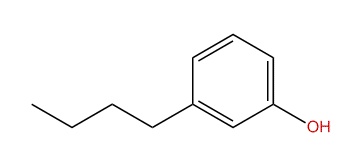
Here the hydroxyl and butyl groups are in a meta position.
Hence the name of the compound is m butyl phenol.
Note:
Finally, if there are three or more substituent classes, the ring is numbered in a manner that assigns the lowest possible numbers to the substituents, as seen in the last row of instances. In the final name, the substituents are classified alphabetically. The numbering refers to the alphabetical order if the substitution is symmetrical (third case from the left).
Complete answer:
Substituted benzene ring compounds have a less systematic nomenclature than alkanes, alkenes, and alkynes. As seen by the combined names mentioned below, a few mono-substituted compounds are named by using a group name as a prefix to "benzene." The rest of these molecules, on the other hand, have one-of-a-kind names. In order to learn these titles, memorization is the only option.
Phenyl, abbreviated Ph-, and benzyl, abbreviated Bn-, are two frequently found substituent groups that incorporate a benzene ring. These are shown along with illustrations of how they should be used. A phenyl group should not be confused with the compound phenol. Ar- for an aryl group is a general and useful standardised notation that complements the use of R- for an alkyl group (any aromatic ring).
Where there are several substituents on a benzene ring, the relative positions of the substituents must be shown by counting the ring carbons or by some other method. The prefixes ortho, meta, and para are generally used to denote a 1,2-, 1,3-, or 1,4- relationship in the case of disubstituted benzenes. The first row of compounds in the following illustrations shows this use in colour. Any disubstituted toluenes have singular names (for example, xylene, cresol, and toluidine), and their isomers are usually labelled with the ortho, meta, or para prefix. Relevant isomers of a few disubstituted benzenes have singular names (e.g. salicylic acid & resorcinol).

Here the hydroxyl and butyl groups are in a meta position.
Hence the name of the compound is m butyl phenol.
Note:
Finally, if there are three or more substituent classes, the ring is numbered in a manner that assigns the lowest possible numbers to the substituents, as seen in the last row of instances. In the final name, the substituents are classified alphabetically. The numbering refers to the alphabetical order if the substitution is symmetrical (third case from the left).
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




