\[Propanoic\;acid \xrightarrow[H_2O]{Cl_2/Red\;P} {\rm{X}}\].
What is \[{\rm{X}}\]?
A) Propanal
B) Propanol
C) propane
D) \[\alpha {\rm{ - }}\] chloro propanoic acid
Answer
618.6k+ views
Hint: We know that the reaction of carboxylic acid which must have \[\alpha {\rm{ - }}\] hydrogen atom with bromine or chlorine results in the formation of \[{\rm{2 - }}\] bromo carboxylic acid or \[{\rm{2 - }}\] chloro carboxylic acid in the presence of red phosphorus. The halogenation takes place during the reaction at the \[\alpha {\rm{ - }}\] carbon atom.
Complete answer:
This reaction is carried out on the basis of Hell - Volhard - Zelinsky halogenation reaction halogenated carboxylic acids at the \[\alpha\] carbon atom.
We know that propanoic acid is the carboxylic acid with \[\alpha {\rm{ - }}\] hydrogen atom. The reaction of propanoic acid with chlorine in the presence of red phosphorus leads to the formation of \[{\rm{2 - }}\]chloropropanoic acid which is also named as \[\alpha {\rm{ - }}\]chloro propanoic acid because the chlorination will take place at \[\alpha {\rm{ - }}\]carbon atom.
We can write the chemical equation for the reaction carried as follows.
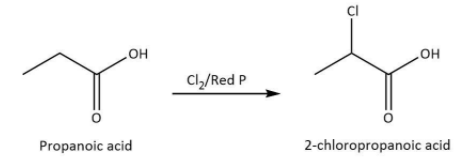
Hence, we can conclude that the correct option is D.
Note: The reaction which is carried out in the above question is commonly known as Hell-Volhard-Zelinsky reaction. The reaction takes place by the formation of phosphorous trichloride as the in-situ which catalysis the reaction. Therefore, we can say that the reaction is acid catalyzed enolization which is followed by the chlorination at alpha position.
Complete answer:
This reaction is carried out on the basis of Hell - Volhard - Zelinsky halogenation reaction halogenated carboxylic acids at the \[\alpha\] carbon atom.
We know that propanoic acid is the carboxylic acid with \[\alpha {\rm{ - }}\] hydrogen atom. The reaction of propanoic acid with chlorine in the presence of red phosphorus leads to the formation of \[{\rm{2 - }}\]chloropropanoic acid which is also named as \[\alpha {\rm{ - }}\]chloro propanoic acid because the chlorination will take place at \[\alpha {\rm{ - }}\]carbon atom.
We can write the chemical equation for the reaction carried as follows.

Hence, we can conclude that the correct option is D.
Note: The reaction which is carried out in the above question is commonly known as Hell-Volhard-Zelinsky reaction. The reaction takes place by the formation of phosphorous trichloride as the in-situ which catalysis the reaction. Therefore, we can say that the reaction is acid catalyzed enolization which is followed by the chlorination at alpha position.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




