Positive carbylamine test is shown by:
A. N, N-dimethylaniline
B. Triethylamine
C. N-methyl aniline
D. P-methyl benzyl amine
Answer
594.9k+ views
Hint:The reagent ${\text{CHC}}{{\text{l}}_3}{\text{ + NaOH}}$ is used to convert the amine functional group into cyanide. The reaction is known as the carbylamine reaction. Only the primary amine gives the carbylamine reaction. The reaction is used to differentiate the primary amine from secondary and tertiary amines. So, we will find the primary amine.
Complete step-by-step solution:The reagent ${\text{CHC}}{{\text{l}}_3}{\text{ + NaOH}}$ converts the amine functional group into cyanide. The conversion of the amine functional group into the isocyanide functional group by the reaction of a primary amine with chloroform and base is known as the Carbylamine reaction. This reaction is used to differentiate amines. So, the carbylamine reaction is known as carbylamine test.
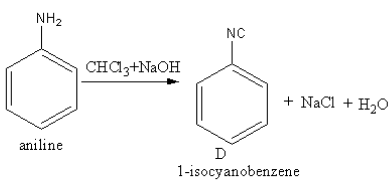
Aniline is a primary amine. The product of the reaction of aniline
With ${\text{CHC}}{{\text{l}}_3}{\text{ + NaOH}}$ is as follows:
Base abstracts the proton from chloroform forming a carbene.The nucleophilic nitrogen of amine attacks the electrophilic carbene. Amine loses one proton and forms a double bond with carbene’s carbon.Again the base abstract the second proton from amine and a triple bond forms between amine’s nitrogen and carbene’s carbon by the removal of chlorine.
The structure of all given amines are as follows:
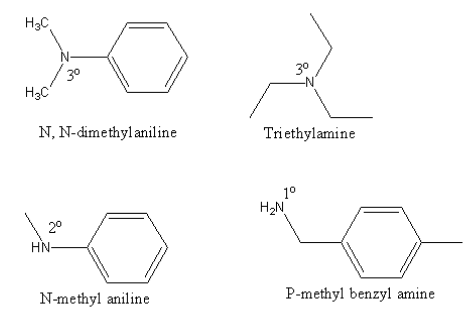
Only the P-methyl benzyl amine is primary amine so it will give the carbylamine test.
Therefore, option (D) P-methyl benzyl amine, is correct.
Note:Reagent used in the Carbylamine reaction is chloroform and a base. The Carbylamine reaction is also known as Hofmann isocyanides synthesis. The presence of two hydrogens is necessary for the carbylamine reaction. The secondary and tertiary amines do not give the carbylamine test. The cyanide group has a specific foul odour. The foul odour indicates the reacting amine is a primary amine.
Complete step-by-step solution:The reagent ${\text{CHC}}{{\text{l}}_3}{\text{ + NaOH}}$ converts the amine functional group into cyanide. The conversion of the amine functional group into the isocyanide functional group by the reaction of a primary amine with chloroform and base is known as the Carbylamine reaction. This reaction is used to differentiate amines. So, the carbylamine reaction is known as carbylamine test.
Aniline is a primary amine. The product of the reaction of aniline
With ${\text{CHC}}{{\text{l}}_3}{\text{ + NaOH}}$ is as follows:
Base abstracts the proton from chloroform forming a carbene.The nucleophilic nitrogen of amine attacks the electrophilic carbene. Amine loses one proton and forms a double bond with carbene’s carbon.Again the base abstract the second proton from amine and a triple bond forms between amine’s nitrogen and carbene’s carbon by the removal of chlorine.

The structure of all given amines are as follows:

Only the P-methyl benzyl amine is primary amine so it will give the carbylamine test.
Therefore, option (D) P-methyl benzyl amine, is correct.
Note:Reagent used in the Carbylamine reaction is chloroform and a base. The Carbylamine reaction is also known as Hofmann isocyanides synthesis. The presence of two hydrogens is necessary for the carbylamine reaction. The secondary and tertiary amines do not give the carbylamine test. The cyanide group has a specific foul odour. The foul odour indicates the reacting amine is a primary amine.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




