\[{{\text{P}}_{4}}{{\text{S}}_{3}}\] has 6 P-S and ____ P-P bonds.
Answer
603.6k+ views
Hint: Phosphorus sesquisulfide, \[{{\text{P}}_{4}}{{\text{S}}_{3}}\], an inorganic compound, with an orthorhombic crystal structure. By studying its geometry, we can determine the number of different types of bonds it has.
Complete Solution :
\[{{\text{P}}_{4}}{{\text{S}}_{3}}\], a yellow solid, is one of the two commercially produced phosphorus sulphides. It is a major component of the strike-anywhere matches. Depending on purity of the compound obtained samples can appear yellow-green to grey. Phosphorus sesquisulphide, free from yellow and white phosphorus appears as a perfect yellow crystalline solid.
- This compound has a trigonal pyramidal geometry, having a triangular base made with phosphorus at each corner of the triangle. Each of the phosphorus on the triangular is perpendicularly attached to a sulphur atom. All these sulphur atoms are further attached to a single trivalent phosphorus atom as shown in the figure given below. It is a derivative of the tetrahedral phosphorus unit from insertion of sulphur into three P-P bonds. The P-S and P-P bond lengths are $2.090\,\overset{\circ }{\mathop{A}}\,$ and $2.235\,\overset{\circ }{\mathop{A}}\,$ respectively.
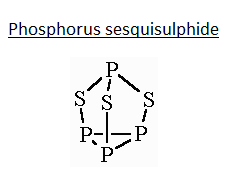
Thus, from the image and explanation given we infer that 6 P-S and 3 P−P bonds are present in the above compound.
Note: \[{{\text{P}}_{4}}{{\text{S}}_{3}}\] compound is made by melting phosphorus and sulphur together at high temperatures and they form the mixed crystals of one dissolved in the other. Excess sulphur gives phosphorus penta-sulphide $({{P}_{4}}{{S}_{10}})$. Its flash point is about ${{100}^{\circ }}C$ .
- It can be easily ignited by friction. It also forms sulphur dioxide and phosphorus penta-oxide during combustion with oxygen. It reacts with water to form phosphoric acid which is a corrosive material. It is also used to make matches and in the manufacture of other chemicals.
Complete Solution :
\[{{\text{P}}_{4}}{{\text{S}}_{3}}\], a yellow solid, is one of the two commercially produced phosphorus sulphides. It is a major component of the strike-anywhere matches. Depending on purity of the compound obtained samples can appear yellow-green to grey. Phosphorus sesquisulphide, free from yellow and white phosphorus appears as a perfect yellow crystalline solid.
- This compound has a trigonal pyramidal geometry, having a triangular base made with phosphorus at each corner of the triangle. Each of the phosphorus on the triangular is perpendicularly attached to a sulphur atom. All these sulphur atoms are further attached to a single trivalent phosphorus atom as shown in the figure given below. It is a derivative of the tetrahedral phosphorus unit from insertion of sulphur into three P-P bonds. The P-S and P-P bond lengths are $2.090\,\overset{\circ }{\mathop{A}}\,$ and $2.235\,\overset{\circ }{\mathop{A}}\,$ respectively.

Thus, from the image and explanation given we infer that 6 P-S and 3 P−P bonds are present in the above compound.
Note: \[{{\text{P}}_{4}}{{\text{S}}_{3}}\] compound is made by melting phosphorus and sulphur together at high temperatures and they form the mixed crystals of one dissolved in the other. Excess sulphur gives phosphorus penta-sulphide $({{P}_{4}}{{S}_{10}})$. Its flash point is about ${{100}^{\circ }}C$ .
- It can be easily ignited by friction. It also forms sulphur dioxide and phosphorus penta-oxide during combustion with oxygen. It reacts with water to form phosphoric acid which is a corrosive material. It is also used to make matches and in the manufacture of other chemicals.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




