On heating glycerol with excess amount of ${\rm{HI}}$, the product formed is:
A. Allyl iodide
B. Isopropyl iodide
C. Propylene
D. $1,2,3 - {\rm{tri iodopropane}}$
Answer
596.1k+ views
Hint: Glycerol has three hydroxyl groups that take part in the reaction and the final product is formed after going through some intermediates. These intermediates decide which type of reaction is going to happen.
Step by step answer: We know that one method of preparing halogenated alkanes is the reaction of alcohols with suitable hydrogen halide. However, we have to take into account the reactant’s properties and reaction conditions as well while deducing the final product. Here, we are given a reaction of heating glycerol with an excess amount of ${\rm{HI}}$. Let’s look at our reactant, first of all which has the following structure:
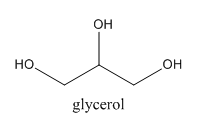
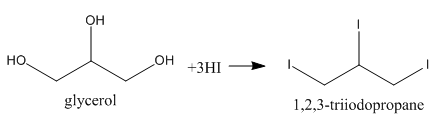
As we can see, three $ - {\rm{OH}}$ groups are present at each of the three carbon atoms. We can write its proper name to be ${\rm{Propane}} - {\rm{1, 2, 3}} - {\rm{triol}}$. Now, upon reacting with excess of ${\rm{HI}}$, we can say that all of the three $ - {\rm{OH}}$ groups would be replaced by iodides as in any normal reaction but here, this product is not stable but an intermediate one. We can show the reaction as follows:
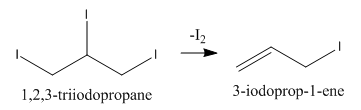
We can write the next step of the reaction in which $1,2,3 - {\rm{triiodopropane}}$ loses a molecule of iodine as follows:
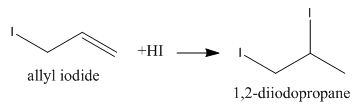
We know that \[{\rm{3 - iodoprop - 1 - ene}}\] is also known as allyl iodide and it can add one molecule of ${\rm{HI}}$ as follows:
As we can see that this addition product, \[{\rm{1,2 - diiodopropane}}\] can also eliminate one iodine molecule as follows:
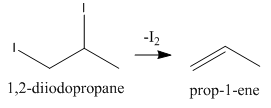
Now, again we have an alkene that can add one molecule of ${\rm{HI}}$ to give the final product as follows:
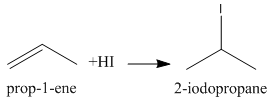
We know that \[{\rm{2 - iodopropane}}\] is also known as Isopropyl iodide.
Hence, the correct option is B.
Note: Here, we have used both the common names as well as IUPAC names for the compounds so it is necessary to know both systems. We have to look for the reaction condition very carefully because the above reaction is possible only when we have an excess amount of ${\rm{HI}}$.
Step by step answer: We know that one method of preparing halogenated alkanes is the reaction of alcohols with suitable hydrogen halide. However, we have to take into account the reactant’s properties and reaction conditions as well while deducing the final product. Here, we are given a reaction of heating glycerol with an excess amount of ${\rm{HI}}$. Let’s look at our reactant, first of all which has the following structure:

As we can see, three $ - {\rm{OH}}$ groups are present at each of the three carbon atoms. We can write its proper name to be ${\rm{Propane}} - {\rm{1, 2, 3}} - {\rm{triol}}$. Now, upon reacting with excess of ${\rm{HI}}$, we can say that all of the three $ - {\rm{OH}}$ groups would be replaced by iodides as in any normal reaction but here, this product is not stable but an intermediate one. We can show the reaction as follows:

We can write the next step of the reaction in which $1,2,3 - {\rm{triiodopropane}}$ loses a molecule of iodine as follows:

We know that \[{\rm{3 - iodoprop - 1 - ene}}\] is also known as allyl iodide and it can add one molecule of ${\rm{HI}}$ as follows:

As we can see that this addition product, \[{\rm{1,2 - diiodopropane}}\] can also eliminate one iodine molecule as follows:

Now, again we have an alkene that can add one molecule of ${\rm{HI}}$ to give the final product as follows:

We know that \[{\rm{2 - iodopropane}}\] is also known as Isopropyl iodide.
Hence, the correct option is B.
Note: Here, we have used both the common names as well as IUPAC names for the compounds so it is necessary to know both systems. We have to look for the reaction condition very carefully because the above reaction is possible only when we have an excess amount of ${\rm{HI}}$.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




