Nylon 6,6 is a ____.
A. Natural polymer
B. Synthetic polymer
C. Mixed polymer
D. Polyester
Answer
543.3k+ views
Hint: A substance consisting of very large molecules or consisting of long chain repeating units are known as polymers. The small units which form the polymers are known as monomers. The polymers are of two types i.e., natural polymer and synthetic polymer.
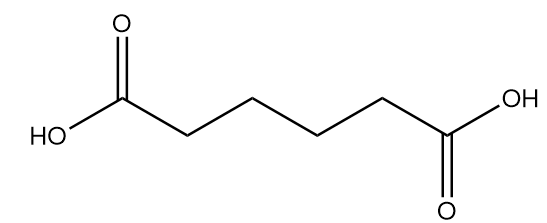
Complete answer: Nylon 6-6 is a synthetic polymer which is also called polyamide. The numbers describe the type and quantity of the polymer chain in the chemical structure. It is composed of two monomers which are hexamethylenediamine and adipic acid. The structures of the monomers of Nylon 66 are as follows:
Adipic acid:
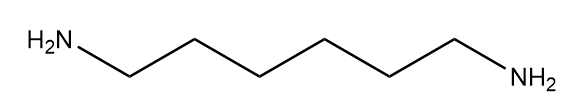
Hexamethylenediamine:
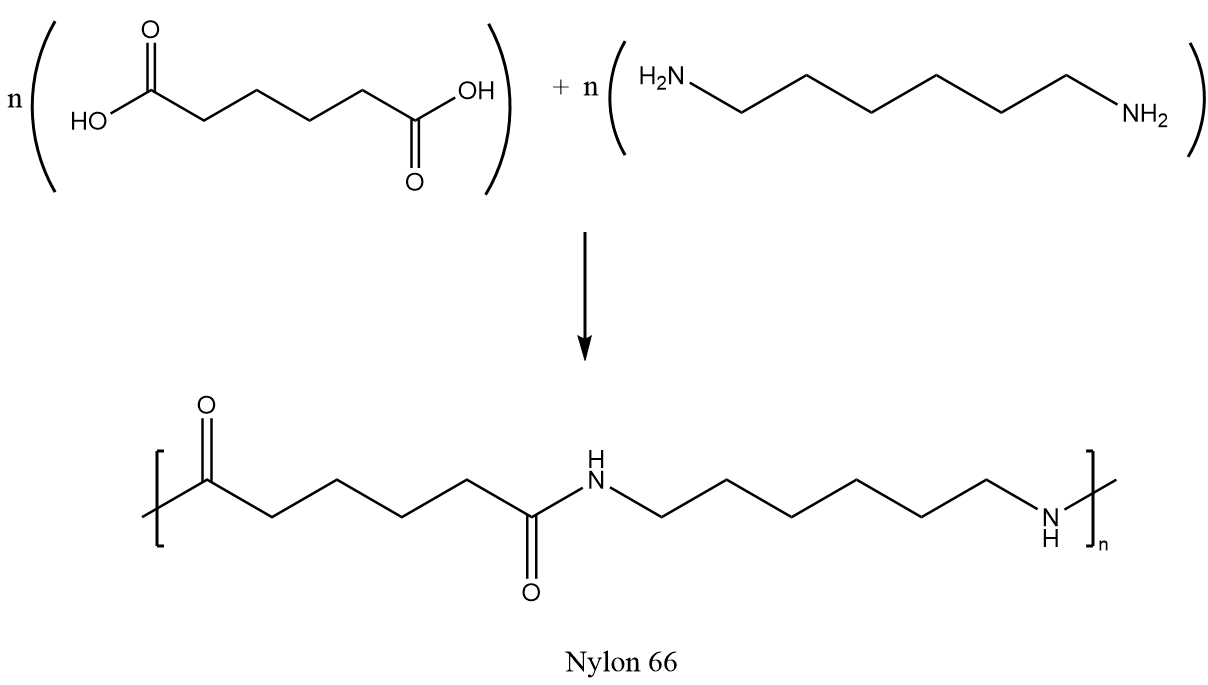
Nylon 6,6 can be synthesized by a polycondensation reaction of adipic acid and hexamethylenediamine followed by the polymerization reaction. The synthesis reaction is as follows:
Hence, Nylon 6,6 is a synthetic polymer.
So, option (B) is the correct answer.
Additional information:
Some important properties of Nylon 6,6 are as follows:
-These polymers have high mechanical strength, hardness, stiffness and toughness.
-These polymers provide good resistance to high radiation energies like gamma and x ray.
-The water absorption rate for these polymers is comparatively lower than other polyamides.
-These polymers have good electrical insulating properties i.e., are good insulators of electricity.
Note:
It is important to note that the major difference between Nylon 6 and Nylon 6,6 polymers is mold shrinkage. The polymer nylon 6 has lower mold shrinkage which adds reliability to stuff whereas in nylon 6,6, due to its greater mold shrinkage, the shape of material or stuff changes by exposing it to cool air.
Complete answer: Nylon 6-6 is a synthetic polymer which is also called polyamide. The numbers describe the type and quantity of the polymer chain in the chemical structure. It is composed of two monomers which are hexamethylenediamine and adipic acid. The structures of the monomers of Nylon 66 are as follows:
Adipic acid:

Hexamethylenediamine:

Nylon 6,6 can be synthesized by a polycondensation reaction of adipic acid and hexamethylenediamine followed by the polymerization reaction. The synthesis reaction is as follows:

Hence, Nylon 6,6 is a synthetic polymer.
So, option (B) is the correct answer.
Additional information:
Some important properties of Nylon 6,6 are as follows:
-These polymers have high mechanical strength, hardness, stiffness and toughness.
-These polymers provide good resistance to high radiation energies like gamma and x ray.
-The water absorption rate for these polymers is comparatively lower than other polyamides.
-These polymers have good electrical insulating properties i.e., are good insulators of electricity.
Note:
It is important to note that the major difference between Nylon 6 and Nylon 6,6 polymers is mold shrinkage. The polymer nylon 6 has lower mold shrinkage which adds reliability to stuff whereas in nylon 6,6, due to its greater mold shrinkage, the shape of material or stuff changes by exposing it to cool air.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




