Name the following compounds.
1.
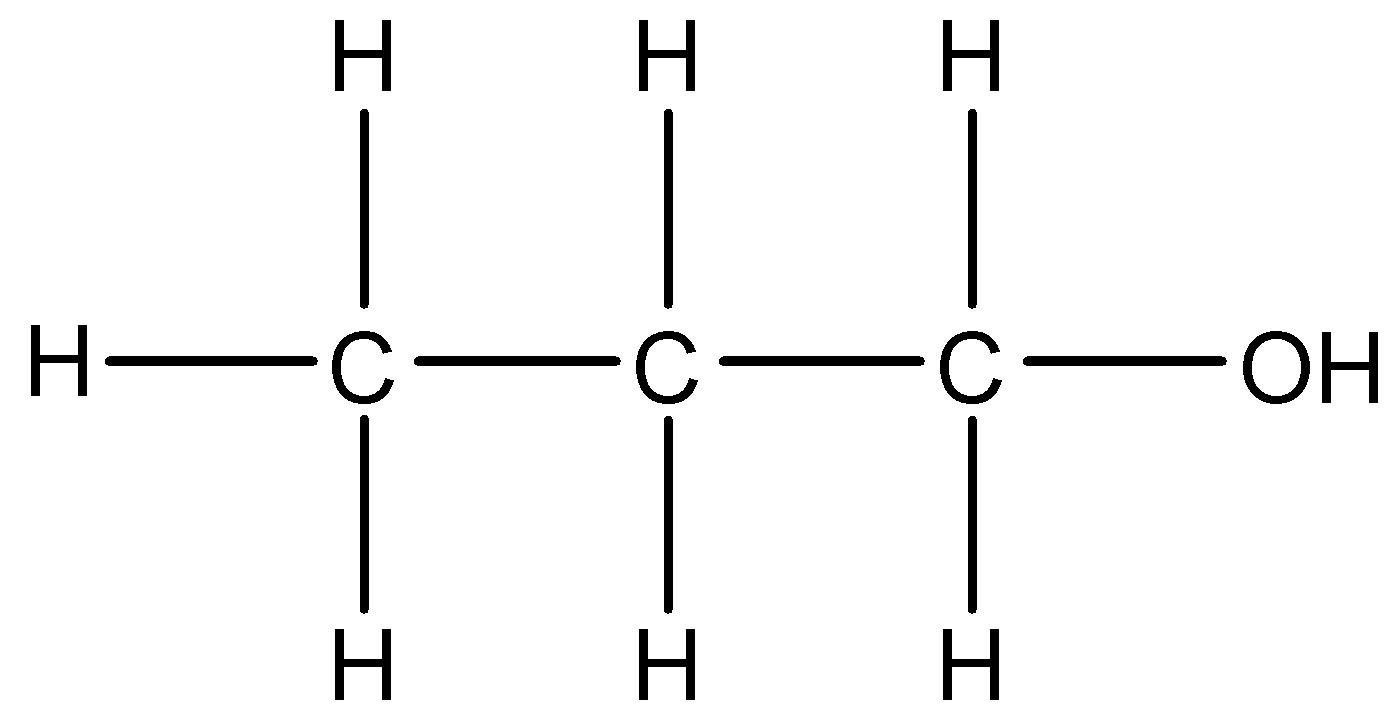
2.
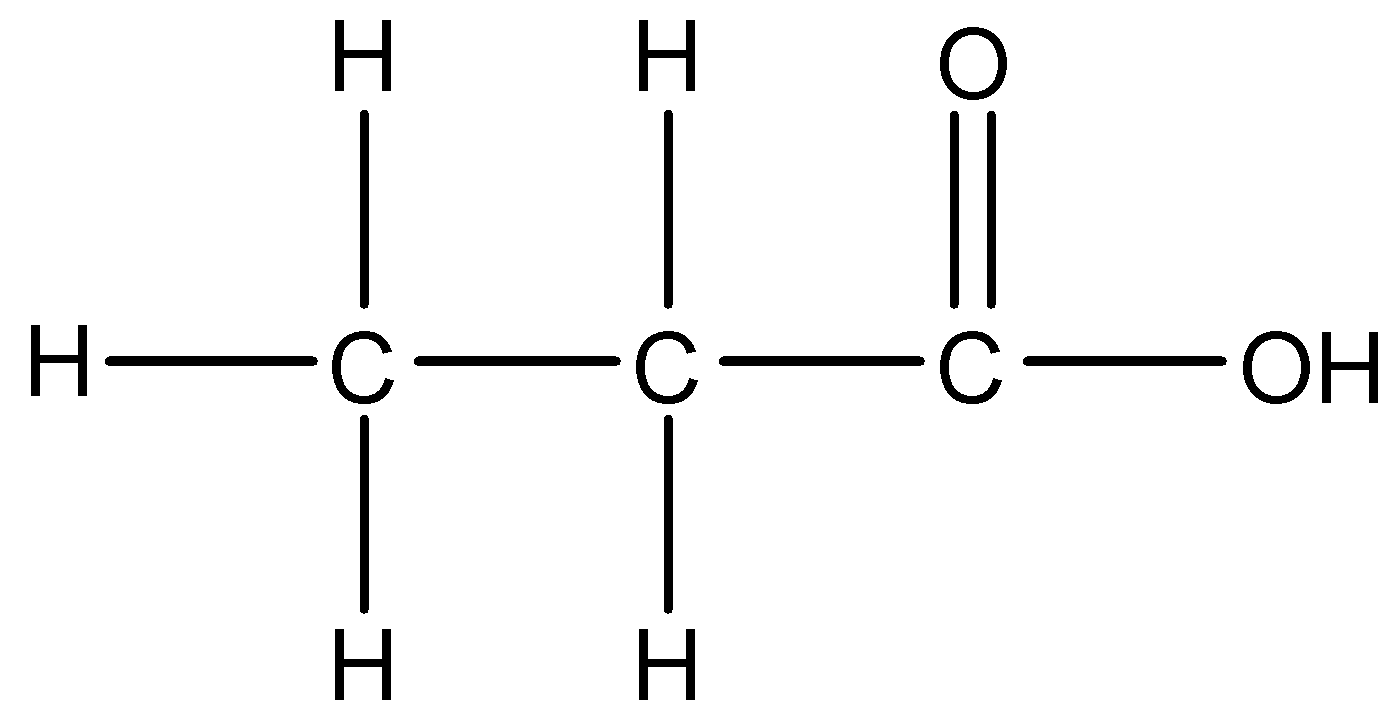
3.
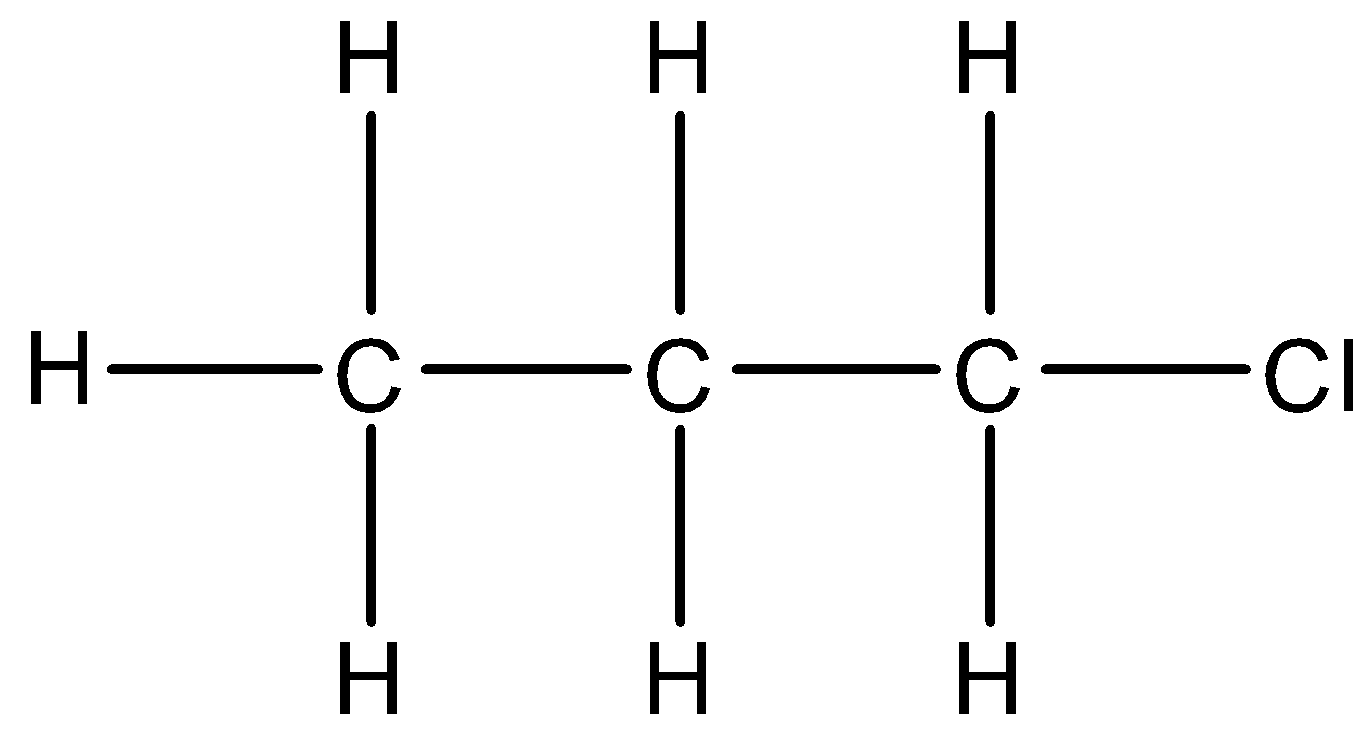
4.
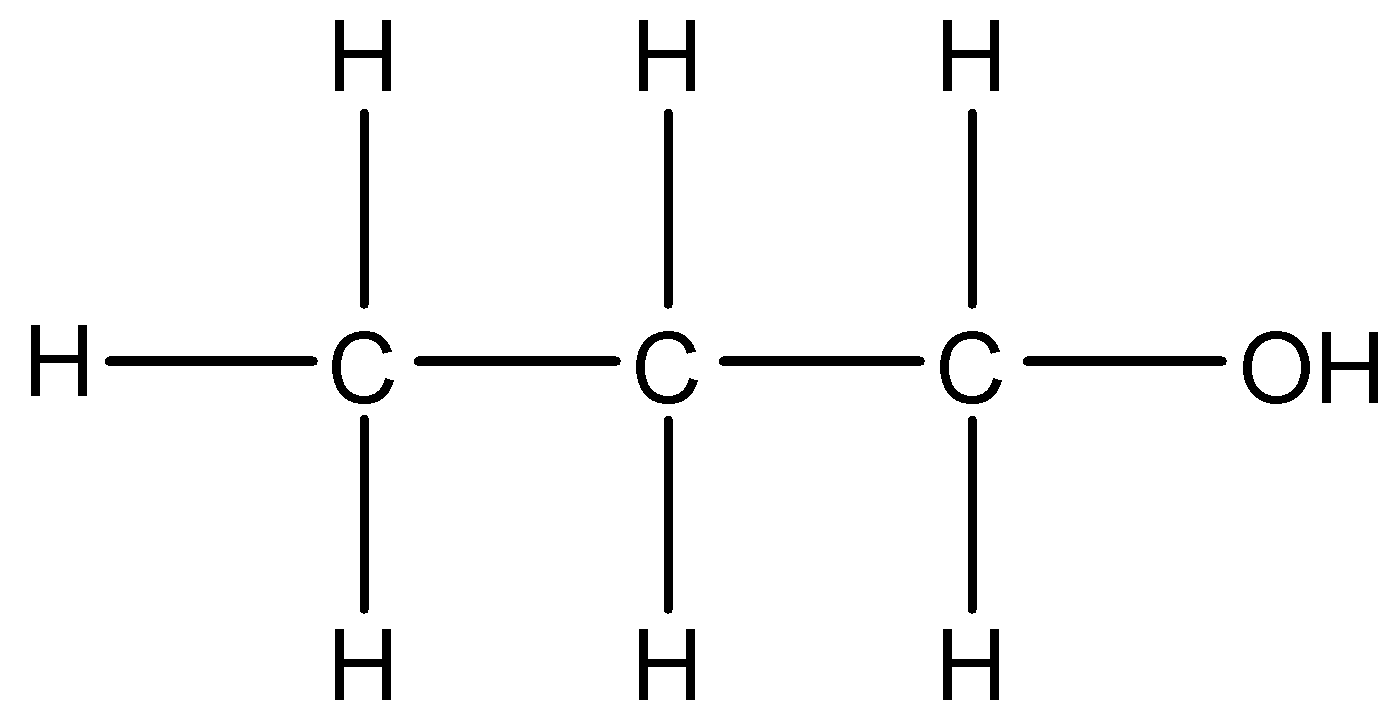
5.
6.






Answer
611.7k+ views
Hint: To name a compound, first find the functional group present in the compound. Then find the longest chain of carbon atoms present in the compound in a way that the carbon of the functional group is involved in the chain.
Complete answer:
We will give the name according to IUPAC nomenclature to all the given compounds one by one.
- To name a compound, first find the functional group present in the compound. Then find the longest chain of carbon atoms present in the compound in a way that the carbon of the functional group is involved in the chain. Then use suitable suffixes of the functional group and indicate the positions of substituent groups as well.
1)
We can see that –OH group is present in the compound. The longest chain of carbon atoms present in the compound is of three carbons. We will use the suffix –ol. Thus, the IUPAC name of this compound will be Propan-1-ol.
2)
There is a –COOH group present in the compound. We know that –oic acid is the suffix used to describe the presence of acids. The parent carbon chain is of three carbons. We will start numbering the chain in a way that the carbon of the functional group gets the lowest number. Thus, the IUPAC name of this compound is 1-Propanoic acid.
3)
There is a chlorine atom in the structure. The parent carbon chain is of three carbons. We will take chlorine atoms as a substituent and use chloro- prefix. So, the IUPAC name of this compound will be 1-chloropropane.
4)
This compound is the same as the first compound.
5)
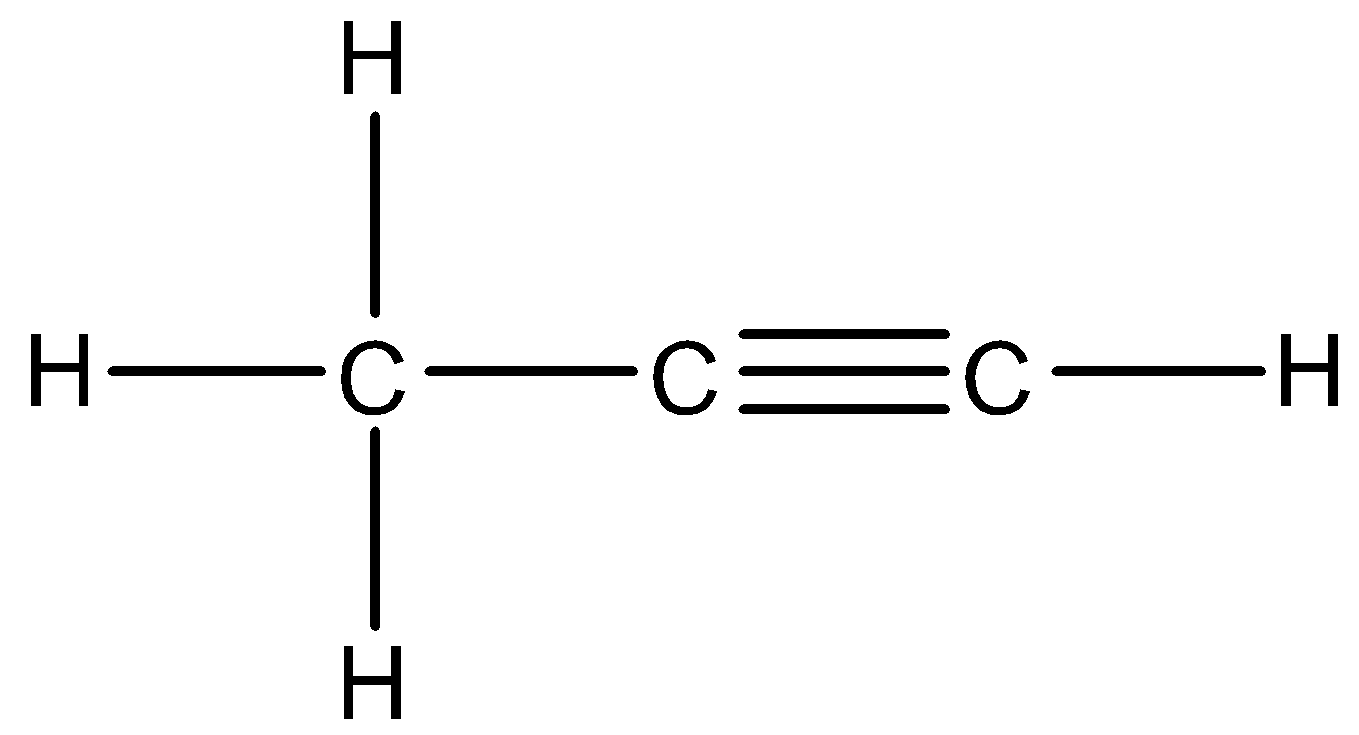
There is a carbon carbon triple bond present in the compound. Thus, this is an alkyne and –yne suffix is used to indicate this functional group. The parent carbon chain consists of three carbons. So, IUPAC name of this compound will be 1-Propyne.
6)
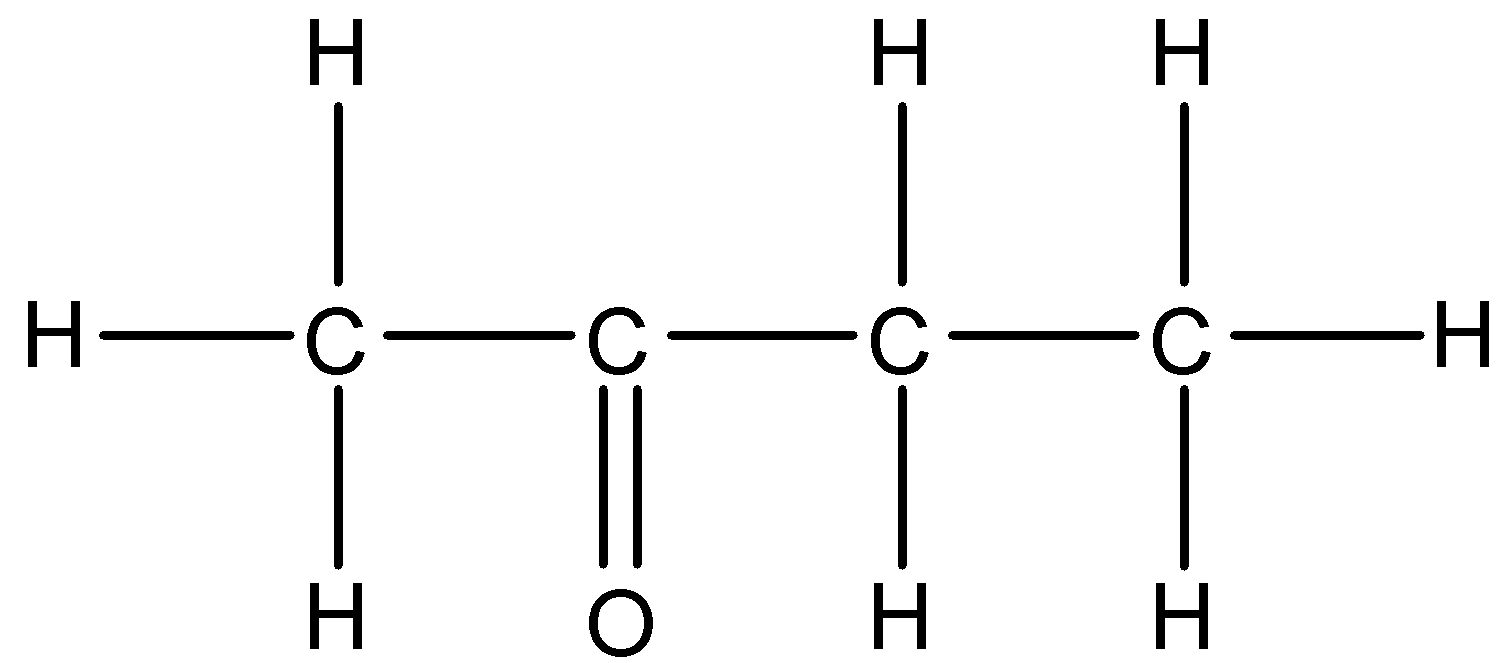
We can see that this compound contains a ketone functional group. So, we will use –one suffix. The parent carbon chain contains four carbon atoms. So, the IUPAC name of this compound will be Butan-2-one.
Note:
When more than one type of functional group is present in the compound, then we need to write them in alphabetical order. If one substituent is present in the compound more than one time, then we can use prefixes like di-, tri-, tetra- etc.
Complete answer:
We will give the name according to IUPAC nomenclature to all the given compounds one by one.
- To name a compound, first find the functional group present in the compound. Then find the longest chain of carbon atoms present in the compound in a way that the carbon of the functional group is involved in the chain. Then use suitable suffixes of the functional group and indicate the positions of substituent groups as well.
1)
We can see that –OH group is present in the compound. The longest chain of carbon atoms present in the compound is of three carbons. We will use the suffix –ol. Thus, the IUPAC name of this compound will be Propan-1-ol.
2)
There is a –COOH group present in the compound. We know that –oic acid is the suffix used to describe the presence of acids. The parent carbon chain is of three carbons. We will start numbering the chain in a way that the carbon of the functional group gets the lowest number. Thus, the IUPAC name of this compound is 1-Propanoic acid.
3)
There is a chlorine atom in the structure. The parent carbon chain is of three carbons. We will take chlorine atoms as a substituent and use chloro- prefix. So, the IUPAC name of this compound will be 1-chloropropane.
4)
This compound is the same as the first compound.
5)
There is a carbon carbon triple bond present in the compound. Thus, this is an alkyne and –yne suffix is used to indicate this functional group. The parent carbon chain consists of three carbons. So, IUPAC name of this compound will be 1-Propyne.
6)
We can see that this compound contains a ketone functional group. So, we will use –one suffix. The parent carbon chain contains four carbon atoms. So, the IUPAC name of this compound will be Butan-2-one.
Note:
When more than one type of functional group is present in the compound, then we need to write them in alphabetical order. If one substituent is present in the compound more than one time, then we can use prefixes like di-, tri-, tetra- etc.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




