Maximum number of electrons present in \[{\text{Ni}}\] for which $\left| {\text{m}} \right| = 1$, is
A) 11
B) 8
C) 12
D) 14
Answer
582k+ views
Hint: We know that an atom consists of electrons. The electrons are small negatively charged particles that move in an orbit around the nucleus. The electrons revolve around the nucleus. A set of numbers which is used to describe the position and movement of an electron in an atom is known as quantum number. There are four quantum numbers namely, principal quantum number, azimuthal quantum number, magnetic quantum number and spin quantum number.
Complete step-by-step answer:
There are four types of quantum numbers:
Principal quantum number: Principal quantum number is designated by $n$. The principal quantum number designates the shell of the atom. Principal quantum number ranges from 1, 2, 3, 4,….
Azimuthal quantum number: Azimuthal quantum number is designated by $l$. The azimuthal quantum number designates the subshell of the atom. If $l = 0$ then it is s-subshell, if $l = 1$ then it is p-subshell, if $l = 2$ then it is d-subshell and if $l = 3$ then it is f-subshell.
Magnetic quantum number: Magnetic quantum number is designated by $m$. The magnetic quantum number designates the total number of orbitals in the subshell and their orientations. The values of magnetic quantum number range between $ + l$ to $ - l$.
Spin quantum number: Spin quantum number is designated by $s$. The spin quantum number designates the spin of the atom. Its values are $ + {\text{1/2}}$ and $ - {\text{1/2}}$.
We are given that the magnetic quantum number is 1.
The magnetic quantum number is always less than or equal to the azimuthal quantum number. Thus, the azimuthal quantum number is less than or equal to 1.
The principal quantum number is always greater than the azimuthal quantum number. Thus, the principal quantum number is greater than or equal to 2.
The atomic number of nickel is 28. The electronic configuration of nickel is as follows:
$1{s^2}\,2{s^2}\,2{p^6}\,3{s^2}\,3{p^6}\,4{s^2}\,3{d^8}$
The principal quantum number must be greater than or equal to 2. Thus, the acceptable atomic orbitals are as follows:
$2s{\text{ }}2p{\text{ }}3s{\text{ }}3p{\text{ }}4s{\text{ }}3d$
The azimuthal quantum number must be greater than or equal to 1. Thus, the acceptable atomic orbitals are as follows:
$2p{\text{ }}3p{\text{ }}3d$
Thus, we have to calculate the maximum number of electrons having $\left| {\text{m}} \right| = 1$ for the electronic configuration of nickel is as follows:
$2{p^6}\,3{p^6}\,3{d^8}$
We are given that $\left| {\text{m}} \right| = 1$. Thus, the magnetic quantum numbers can be $ + 1{\text{ and }} - 1$. Thus, for $2p{\text{ }}3p{\text{ }}3d$
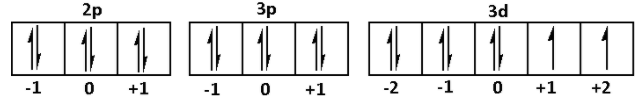
From the figure, e maximum number of electrons having $\left| {\text{m}} \right| = 1$ are 11.
Thus, maximum number of electrons present in \[{\text{Ni}}\] for which $\left| {\text{m}} \right| = 1$ is 11.
Thus, the correct answer is option ‘A’.
Note: Remember for p-orbital the values of magnetic quantum number are $ - 1,0, + 1$ and for the d-orbital the values of magnetic quantum number are $ - 2, - 1,0, + 1, + 2$. We are given that $\left| {\text{m}} \right| = 1$. This indicates the value of magnetic quantum numbers can be positive or negative.
Complete step-by-step answer:
There are four types of quantum numbers:
Principal quantum number: Principal quantum number is designated by $n$. The principal quantum number designates the shell of the atom. Principal quantum number ranges from 1, 2, 3, 4,….
Azimuthal quantum number: Azimuthal quantum number is designated by $l$. The azimuthal quantum number designates the subshell of the atom. If $l = 0$ then it is s-subshell, if $l = 1$ then it is p-subshell, if $l = 2$ then it is d-subshell and if $l = 3$ then it is f-subshell.
Magnetic quantum number: Magnetic quantum number is designated by $m$. The magnetic quantum number designates the total number of orbitals in the subshell and their orientations. The values of magnetic quantum number range between $ + l$ to $ - l$.
Spin quantum number: Spin quantum number is designated by $s$. The spin quantum number designates the spin of the atom. Its values are $ + {\text{1/2}}$ and $ - {\text{1/2}}$.
We are given that the magnetic quantum number is 1.
The magnetic quantum number is always less than or equal to the azimuthal quantum number. Thus, the azimuthal quantum number is less than or equal to 1.
The principal quantum number is always greater than the azimuthal quantum number. Thus, the principal quantum number is greater than or equal to 2.
The atomic number of nickel is 28. The electronic configuration of nickel is as follows:
$1{s^2}\,2{s^2}\,2{p^6}\,3{s^2}\,3{p^6}\,4{s^2}\,3{d^8}$
The principal quantum number must be greater than or equal to 2. Thus, the acceptable atomic orbitals are as follows:
$2s{\text{ }}2p{\text{ }}3s{\text{ }}3p{\text{ }}4s{\text{ }}3d$
The azimuthal quantum number must be greater than or equal to 1. Thus, the acceptable atomic orbitals are as follows:
$2p{\text{ }}3p{\text{ }}3d$
Thus, we have to calculate the maximum number of electrons having $\left| {\text{m}} \right| = 1$ for the electronic configuration of nickel is as follows:
$2{p^6}\,3{p^6}\,3{d^8}$
We are given that $\left| {\text{m}} \right| = 1$. Thus, the magnetic quantum numbers can be $ + 1{\text{ and }} - 1$. Thus, for $2p{\text{ }}3p{\text{ }}3d$

From the figure, e maximum number of electrons having $\left| {\text{m}} \right| = 1$ are 11.
Thus, maximum number of electrons present in \[{\text{Ni}}\] for which $\left| {\text{m}} \right| = 1$ is 11.
Thus, the correct answer is option ‘A’.
Note: Remember for p-orbital the values of magnetic quantum number are $ - 1,0, + 1$ and for the d-orbital the values of magnetic quantum number are $ - 2, - 1,0, + 1, + 2$. We are given that $\left| {\text{m}} \right| = 1$. This indicates the value of magnetic quantum numbers can be positive or negative.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




