What is the major organic compound formed in the reaction of 1-pentene with diborane in diglyme, followed by basic hydrogen peroxide.
(A) 1-pentanol
(B) 2-pentanol
(C) 3-pentanol
(D) None of these
Answer
584.1k+ views
Hint: Write the expanded structure of 1-pentene to identify the position of double bond. Now try to understand the reagents used in the following reaction. The above reaction leads to the formation of Hoffmann product due to the presence of peroxide initiator. Identify the attacking nucleophile in order to arrive at the final product.
Complete step by step answer:
- So in the question it is asked to predict the product formed from 1-pentene, if it is treated with diborane in diglyme, followed by basic hydrogen peroxide
- Hence to solve the question, first we will draw the expanded structure of 1-pentene as suggested in the hint.
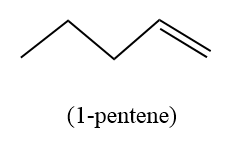
- Diborane in diglyme, followed by basic hydrogen peroxide is used to add hydroxide ion across the double bond in the organic compound. However, it does not follow the Saytzeff rule due to the presence of peroxide.
- The alkanol thus formed is in accordance with the Hoffmann rule and the product formed is called Hofmann product.
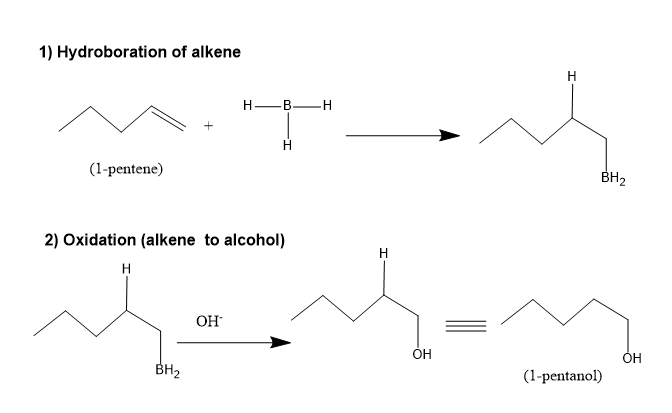
- This reaction is the hydroboration –oxidation reaction of the alkenes and hence the reaction takes place in two steps to yield an alcohol from alkene molecule.
- The first step will be the hydroboration reaction in which the alkene reacts with the borane molecule, and then it is followed by oxidation in the presence of hydrogen peroxide and a base to yield the alcohol.
The two steps that comprises this reaction is mentioned below:
Hence there reaction can be written as,
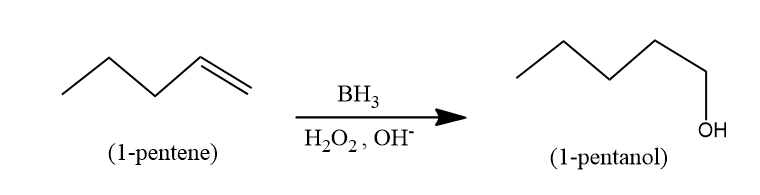
Therefore, the major organic product formed is 1-pentanol.
The correct answer is option “A” .
Note: Do not confuse Hoffmann's rule with Hofmann rearrangement. Hoffmann rearrangement is an organic reaction of a primary amide leading to the formation of a primary amine with one fewer carbon atom.
- Along with this exists one more reaction i.e. the Hofmann elimination. This elimination reaction is for an anime where the least stable alkene is formed. The Hoffmann reaction series has a reaction mechanism opposite to the series of reactions based on Saytzeff rule. All Hoffmann reactions involve the use of peroxide as an initiator.
Complete step by step answer:
- So in the question it is asked to predict the product formed from 1-pentene, if it is treated with diborane in diglyme, followed by basic hydrogen peroxide
- Hence to solve the question, first we will draw the expanded structure of 1-pentene as suggested in the hint.

- Diborane in diglyme, followed by basic hydrogen peroxide is used to add hydroxide ion across the double bond in the organic compound. However, it does not follow the Saytzeff rule due to the presence of peroxide.
- The alkanol thus formed is in accordance with the Hoffmann rule and the product formed is called Hofmann product.
- This reaction is the hydroboration –oxidation reaction of the alkenes and hence the reaction takes place in two steps to yield an alcohol from alkene molecule.
- The first step will be the hydroboration reaction in which the alkene reacts with the borane molecule, and then it is followed by oxidation in the presence of hydrogen peroxide and a base to yield the alcohol.
The two steps that comprises this reaction is mentioned below:

Hence there reaction can be written as,

Therefore, the major organic product formed is 1-pentanol.
The correct answer is option “A” .
Note: Do not confuse Hoffmann's rule with Hofmann rearrangement. Hoffmann rearrangement is an organic reaction of a primary amide leading to the formation of a primary amine with one fewer carbon atom.
- Along with this exists one more reaction i.e. the Hofmann elimination. This elimination reaction is for an anime where the least stable alkene is formed. The Hoffmann reaction series has a reaction mechanism opposite to the series of reactions based on Saytzeff rule. All Hoffmann reactions involve the use of peroxide as an initiator.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




