Lithium forms body-centered cubic structure. The length of the side of its unit cell is 351 pm. Atomic radius of lithium will be:
A. 75pm
B. 300pm
C. 240pm
D. 152pm
Answer
602.4k+ views
Hint: In a body centered unit length the radius and side length are related by the equation \[r=(\surd 3\div 4)\times a\], where a is the edge length of the unit cell. Use this equation to derive the atomic radius of the element.
Complete step-by step solution:
In order to understand the question, let us first understand the structure of a body-centered cubic unit cell. The diagram is given below:
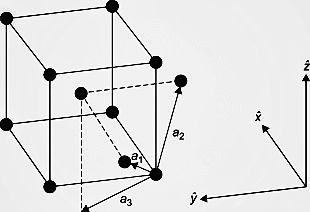
-As we can see from the figure, in BCC, there are 8 atoms at the 8 edges of the cube and one atom is present at the centre of the cube. All of these contribute to the BCC structure and let us find their individual contributions.
-As there are 8 edges in a cube and all of them are filled with 8 atoms, then we can say that each atom in the edge has a contribution of 1 8 to the BCC structure. There is a single atom present at the centre of the cube, so it is counted as 1 i.e full contribution to the cell.
-Hence, the total contribution of BCC cell is \[\]\[(1\times (1\div 8))from\text{ }edges+1\text{ }from\text{ }body\text{ }centre=\text{ }2\text{ }atoms\].
-Also, the body diagonal has a length that is 4 times the radius of the atom,r. Using Pythagoras theorem, we get the relation as
\[r=(\surd 3\div 4)\times a\]
-Substituting the value of a as 351 pm, we have
$r=\dfrac{\sqrt{3}}{4} \times 351 pm = 152pm$
So we obtain the radius of the lithium atom as 152pm.,
Note: As the question does not contain SI units and the answer too, it is not at all required to change pico metre to metre. It will consume more time. Take √3 to be approximately 1.73. Also remember that the packing fraction of BCC is about 68%.
Complete step-by step solution:
In order to understand the question, let us first understand the structure of a body-centered cubic unit cell. The diagram is given below:

-As we can see from the figure, in BCC, there are 8 atoms at the 8 edges of the cube and one atom is present at the centre of the cube. All of these contribute to the BCC structure and let us find their individual contributions.
-As there are 8 edges in a cube and all of them are filled with 8 atoms, then we can say that each atom in the edge has a contribution of 1 8 to the BCC structure. There is a single atom present at the centre of the cube, so it is counted as 1 i.e full contribution to the cell.
-Hence, the total contribution of BCC cell is \[\]\[(1\times (1\div 8))from\text{ }edges+1\text{ }from\text{ }body\text{ }centre=\text{ }2\text{ }atoms\].
-Also, the body diagonal has a length that is 4 times the radius of the atom,r. Using Pythagoras theorem, we get the relation as
\[r=(\surd 3\div 4)\times a\]
-Substituting the value of a as 351 pm, we have
$r=\dfrac{\sqrt{3}}{4} \times 351 pm = 152pm$
So we obtain the radius of the lithium atom as 152pm.,
Note: As the question does not contain SI units and the answer too, it is not at all required to change pico metre to metre. It will consume more time. Take √3 to be approximately 1.73. Also remember that the packing fraction of BCC is about 68%.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




