List the following carbocation in order of decreasing stabilization energies:
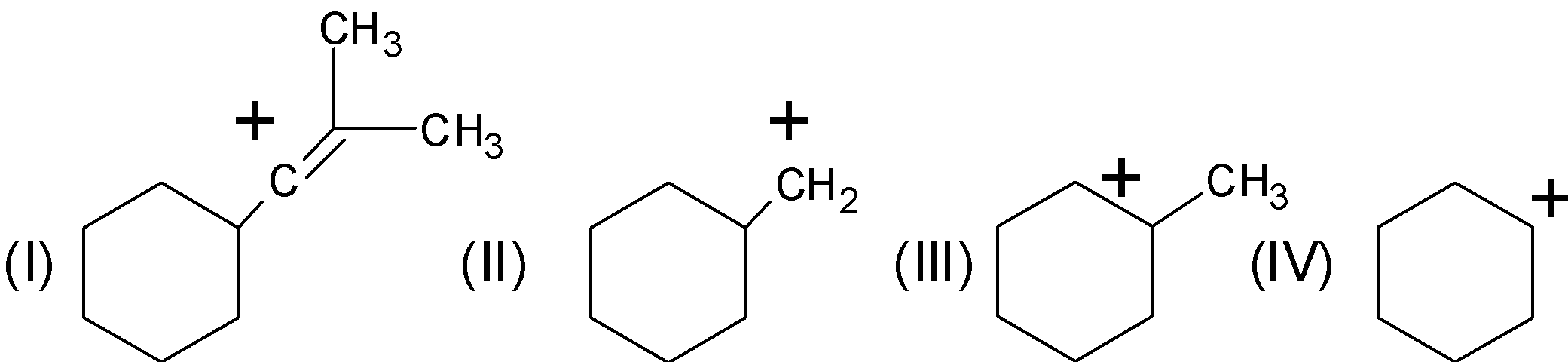
A) $\text{II , III , I , IV}$
B) $\text{III , IV , II , I}$
C) $\text{III , IV , I , III}$
D) $\text{I , II , IV , III}$

Answer
621.3k+ views
Hint: Hyper conjugation is used to depict the stability of the compound. The stability of the carbocation can be measured by the total number of $\text{ }\!\!\alpha\!\!\text{ }$ hydrogen atoms concerning the carbon atom bearing a positive charge. If the structures have the same number of $\text{ }\!\!\alpha\!\!\text{ }$hydrogen atoms then go for the hybridization of the carbocation.
Complete step by step answer:
-The stability of carbocation is related to the energy for a stabilization. More stable carbocation, the less is the stabilization energy.
-Here, we will use the concept of hyperconjugation to determine the stability of carbocation.
-Hyper conjugation is called the stabilization interaction. It arises due to the interaction of electrons in $\sigma $ bond (usually these are $\text{C-H or C-C}$ bonds) with the empty or partially filled p-orbital. This gives the extended molecular structure and increases the stability of the system.
-According to the hyper conjugation, the stability increases with the number of $\alpha -H$ atoms bonded to the carbon forming carbocation. Thus more the number of $\alpha -H$atoms greater is the stability and less is the stabilization energy required.
-Let us have a look at the option.
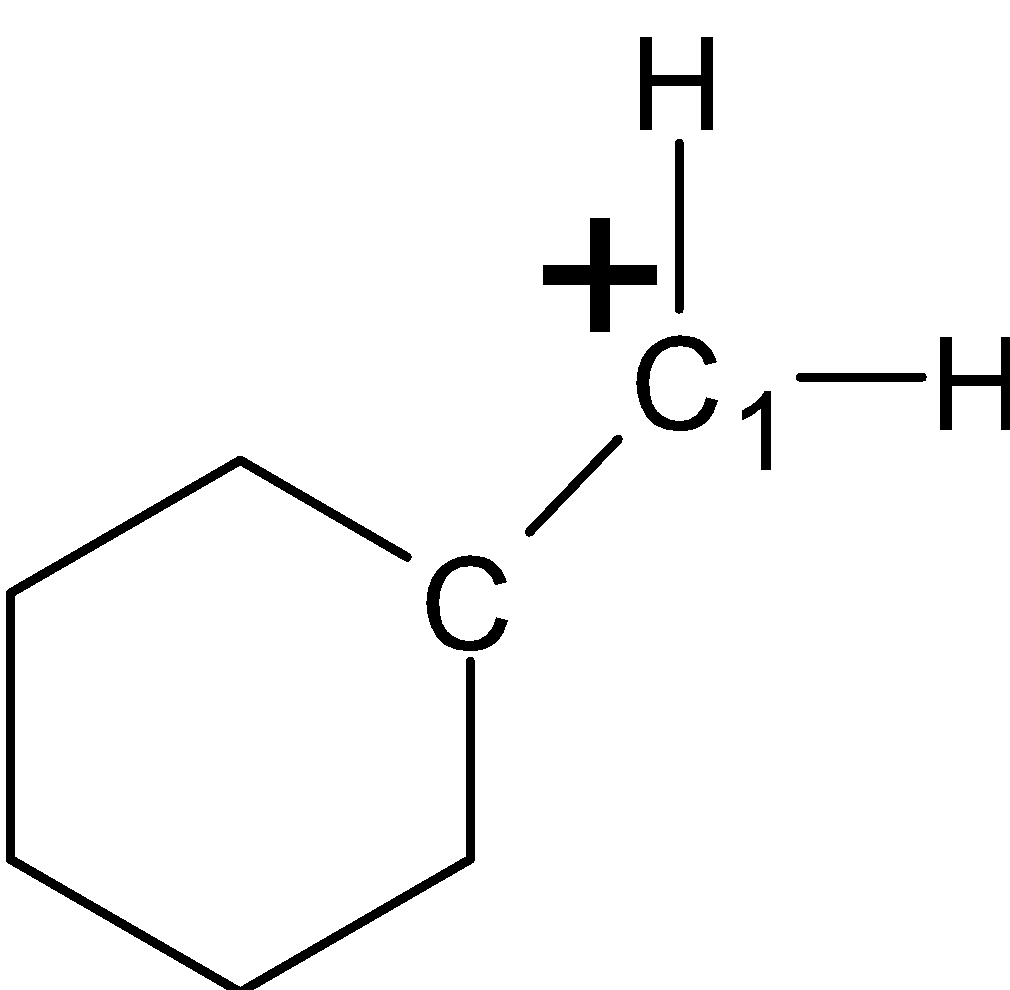
-Here, in the structure (I) the carbocation is formed at the double-bonded carbon${{\text{C}}_{\text{2}}}$. The ${{\text{C}}_{\text{2}}}$carbon is bonded to the one $\alpha -H$atom at the $\text{C}$ring. The $\alpha -H$ as shown below.
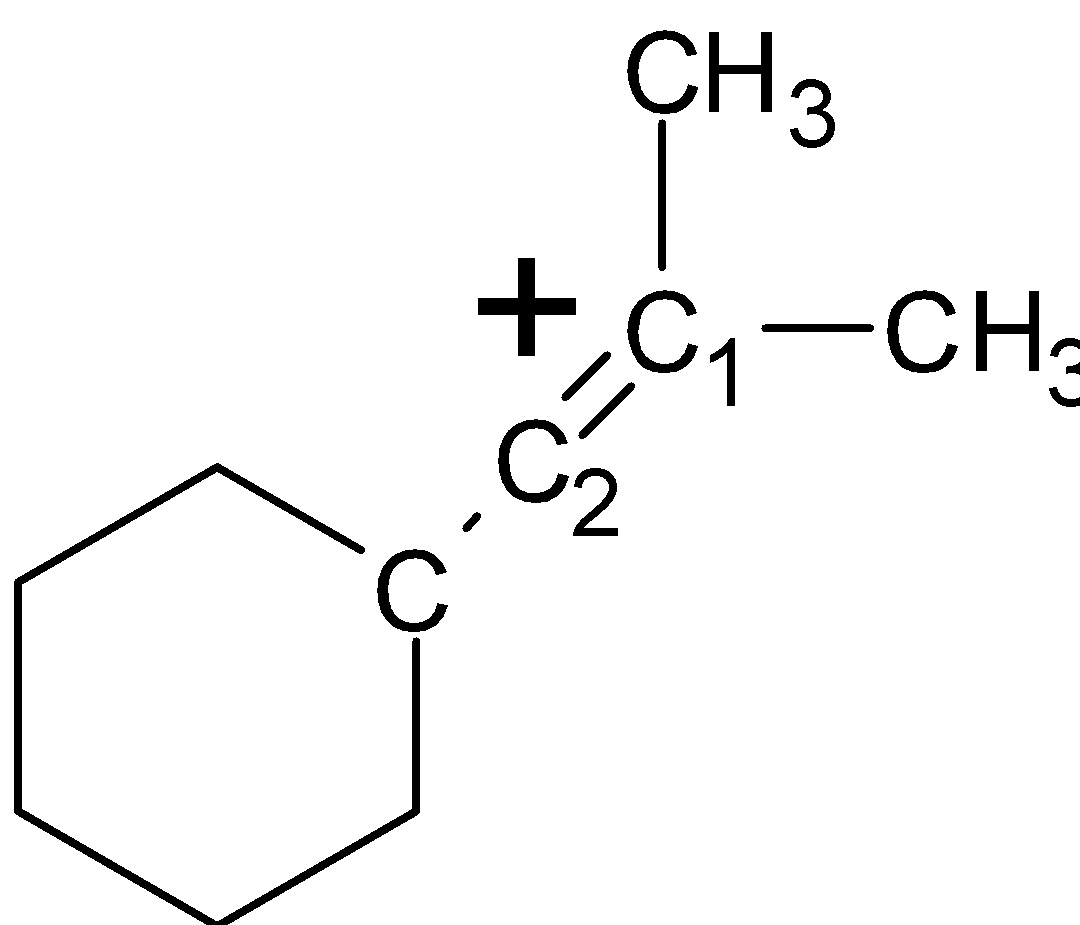
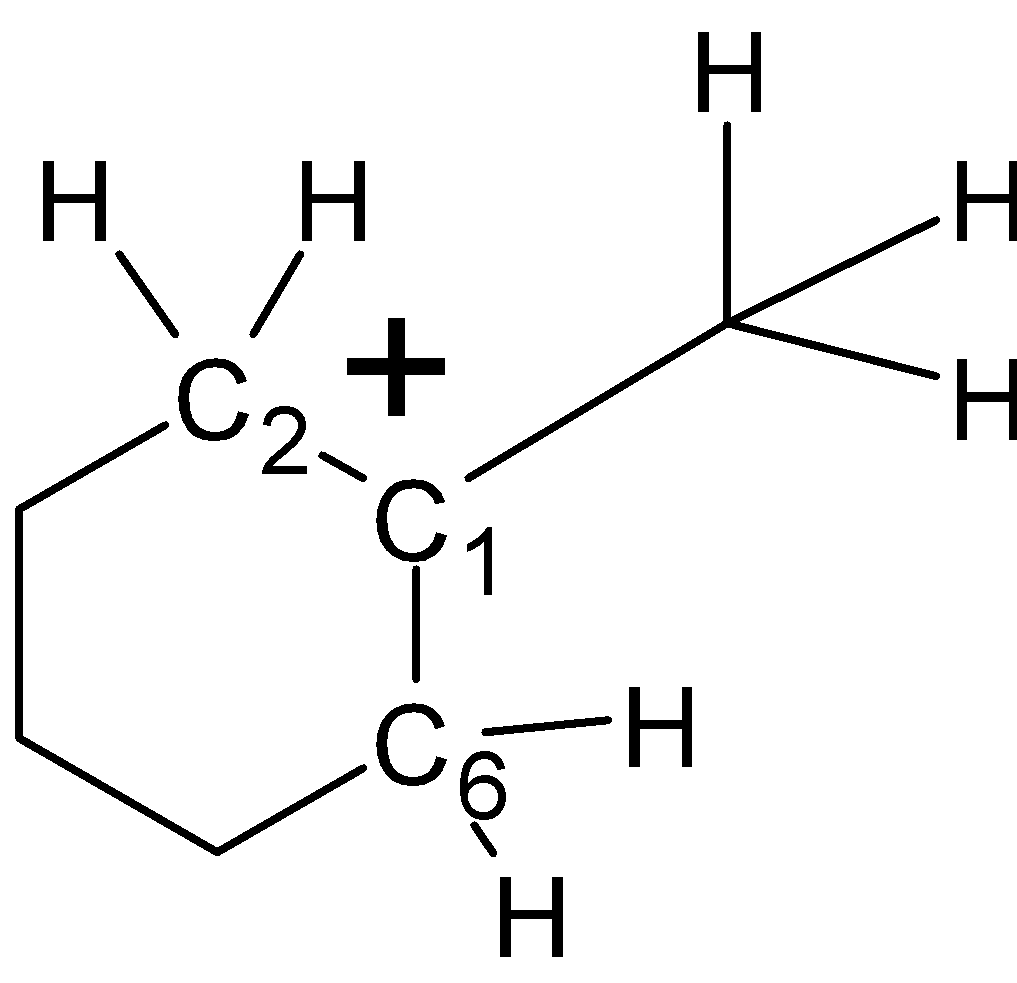
-In structure (III), the carbocation is formed at the ${{\text{C}}_{1}}$ carbon as shown in the structure. There is a total of four $\alpha -H$atoms at the ${{\text{C}}_{\text{2}}}$and ${{\text{C}}_{6}}$ position of the ring. Along with that, there are three more $\alpha -H$atoms at the ${{\text{C}}_{1}}$ carbon atom. Therefore there is a total of 7 $\alpha -H$atoms. As shown below,
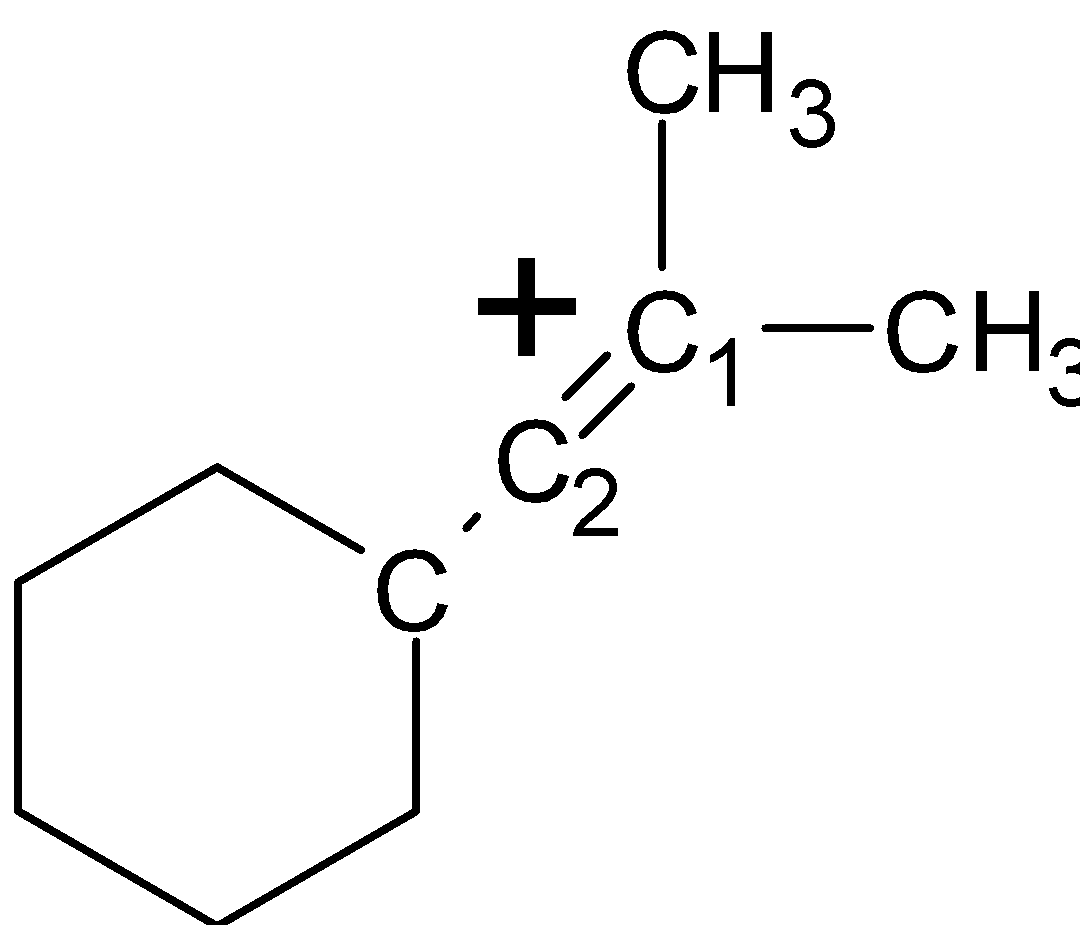
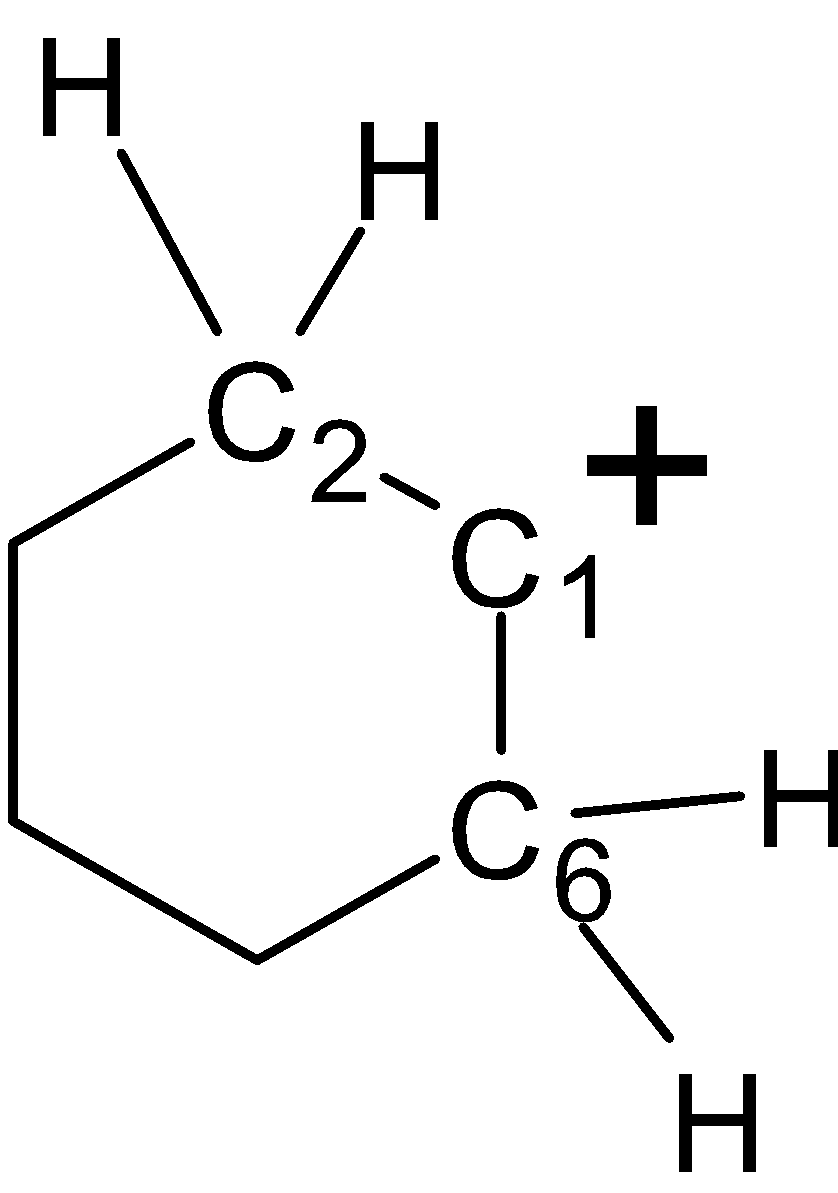
-In structure (IV), let the carbocation is at the ring carbon atom. There are two $\alpha -H$atoms at the ${{\text{C}}_{\text{2}}}$ ring and two at the ${{\text{C}}_{6}}$of the ring .the ${{\text{C}}_{\text{2}}}$and ${{\text{C}}_{6}}$ carbon atoms are at the $\alpha $ to the ${{\text{C}}_{1}}$ carbon. Thus the structure (III) has 4 $\alpha -H$atoms. The structure as shown below,
-Thus, the structure (III) is the most stable carbocation followed by the (IV) structure. However, there is still an ambiguity among the structure (I) and (II) as both have the same number of $\alpha -H$ atoms.
Therefore, let us consider the hybridization of the carbon atom. In structure (I), the carbocation is $\text{s}{{\text{p}}^{\text{2}}}$ hybridized which is highly unstable. Therefore (I) will be the least stable of all structures.
Therefore the decreasing order of stability or the stabilization energy is:
$\text{IIIIVIII}$
Hence, (B) is the correct option.
Note: The stability of carbocation also increases as we move from primary to secondary to a tertiary carbon atom.
$\text{Primary (}{{\text{1}}^{\text{0}}}\text{) Secondary (}{{\text{2}}^{\text{0}}}\text{) Tertiary (}{{\text{3}}^{\text{0}}}\text{)}$
Complete step by step answer:
-The stability of carbocation is related to the energy for a stabilization. More stable carbocation, the less is the stabilization energy.
-Here, we will use the concept of hyperconjugation to determine the stability of carbocation.
-Hyper conjugation is called the stabilization interaction. It arises due to the interaction of electrons in $\sigma $ bond (usually these are $\text{C-H or C-C}$ bonds) with the empty or partially filled p-orbital. This gives the extended molecular structure and increases the stability of the system.
-According to the hyper conjugation, the stability increases with the number of $\alpha -H$ atoms bonded to the carbon forming carbocation. Thus more the number of $\alpha -H$atoms greater is the stability and less is the stabilization energy required.
-Let us have a look at the option.
-Here, in the structure (I) the carbocation is formed at the double-bonded carbon${{\text{C}}_{\text{2}}}$. The ${{\text{C}}_{\text{2}}}$carbon is bonded to the one $\alpha -H$atom at the $\text{C}$ring. The $\alpha -H$ as shown below.



-In structure (III), the carbocation is formed at the ${{\text{C}}_{1}}$ carbon as shown in the structure. There is a total of four $\alpha -H$atoms at the ${{\text{C}}_{\text{2}}}$and ${{\text{C}}_{6}}$ position of the ring. Along with that, there are three more $\alpha -H$atoms at the ${{\text{C}}_{1}}$ carbon atom. Therefore there is a total of 7 $\alpha -H$atoms. As shown below,

-In structure (IV), let the carbocation is at the ring carbon atom. There are two $\alpha -H$atoms at the ${{\text{C}}_{\text{2}}}$ ring and two at the ${{\text{C}}_{6}}$of the ring .the ${{\text{C}}_{\text{2}}}$and ${{\text{C}}_{6}}$ carbon atoms are at the $\alpha $ to the ${{\text{C}}_{1}}$ carbon. Thus the structure (III) has 4 $\alpha -H$atoms. The structure as shown below,

-Thus, the structure (III) is the most stable carbocation followed by the (IV) structure. However, there is still an ambiguity among the structure (I) and (II) as both have the same number of $\alpha -H$ atoms.
Therefore, let us consider the hybridization of the carbon atom. In structure (I), the carbocation is $\text{s}{{\text{p}}^{\text{2}}}$ hybridized which is highly unstable. Therefore (I) will be the least stable of all structures.
Therefore the decreasing order of stability or the stabilization energy is:
$\text{IIIIVIII}$
Hence, (B) is the correct option.
Note: The stability of carbocation also increases as we move from primary to secondary to a tertiary carbon atom.
$\text{Primary (}{{\text{1}}^{\text{0}}}\text{) Secondary (}{{\text{2}}^{\text{0}}}\text{) Tertiary (}{{\text{3}}^{\text{0}}}\text{)}$
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




