l-chorobutane on reaction with alcoholic potash gives:
(A) 1-butene
(B) 1-butanol
(C) 2-butane
(D) 2-butanol
Answer
609k+ views
Hint: In alkenes, the molecule contains the double bond between the adjacent carbon atoms. The alkenes can be synthesized from the haloalkanes. Haloalkanes are treated with the potassium hydroxide $\text{ KOH }$. The $\text{ }\beta \text{ }$ hydrogen atom shifts its bonds and forms the double bond followed by the removal of the halogen atom at the $\alpha $ position. The reaction contains the removal of hydrogen and halogen, thus the reaction is known as dehydrohalogenation.
Complete step by step answer:
Let us first start the answer by writing the chemical reaction. So the chemical reaction goes as follows:
\[\text{C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{2}}}\text{Cl }\!\!~\!\!\text{ }\xrightarrow{\text{alcoholic KOH}}\text{ C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\text{CH = C}{{\text{H}}_{\text{2}}}\]
In the above reaction, alcoholic KOH plays the role of a dehydrohalogenation agent. The alcoholic KOH reacts with an alkyl halide and an alkene is formed as the product of the chemical reaction.
The product that is formed consists of 4 carbon atoms and as we can see there is a double bond that is present between the first and the second carbon atoms. So, the name of the compound will be 1-butene.
The mechanism is as shown below:
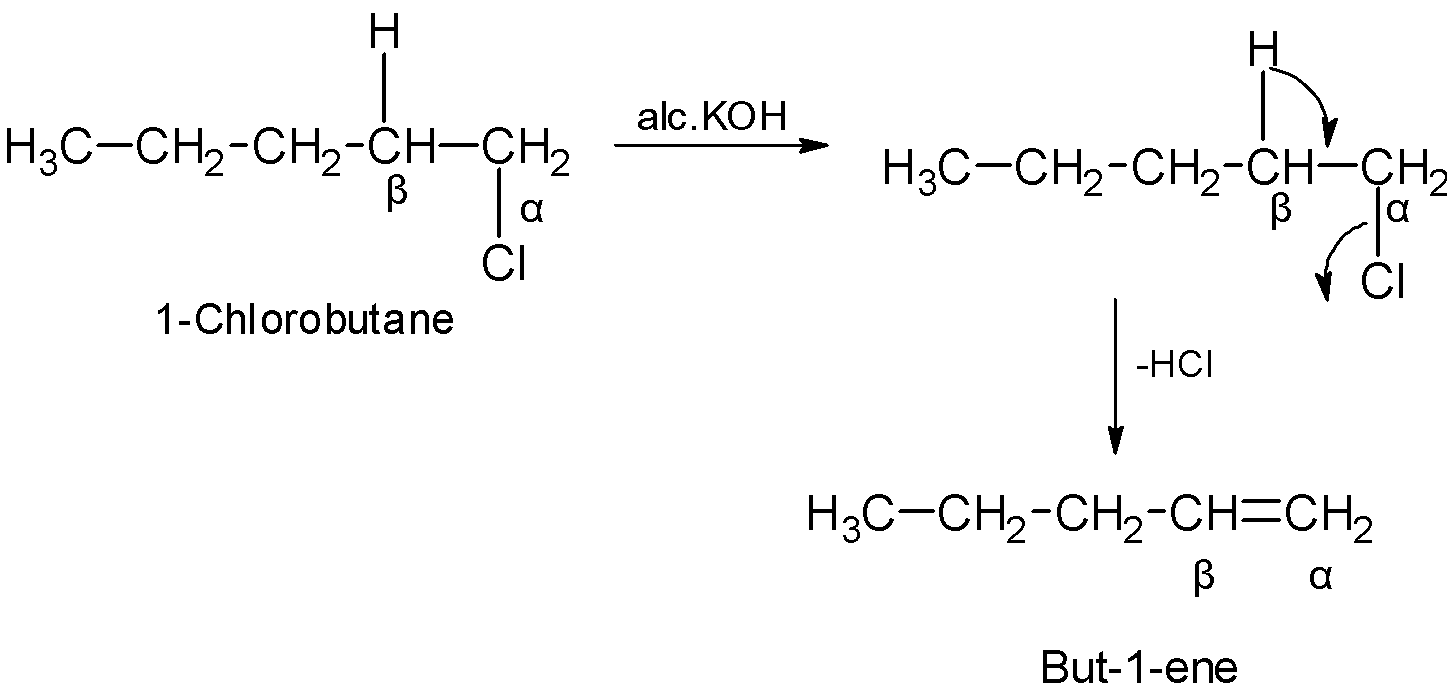
The answer to the question which is 1-butene is an organic compound that is highly flammable and is a highly condensed gas. 1-butene is soluble in ether and alcohol but is not soluble in water.
So, the correct answer is “Option A”.
Additional Information:
By alcoholic solution, as mentioned in the answer, we mean a solution which is a mixture of water and ethanol, which is used as a solvent. Substances that contain sugar can be fermented using this alcoholic solution.
Note: Alcoholic potash that is used in the chemical reaction mentioned above is a very strong base. Alcoholic potash or as we know KOH dissolves in water to give $\text{R}{{\text{O}}^{-}}$ ions. Alcohol abstracts the hydrogen which results in the formation of elimination in the reaction.
Complete step by step answer:
Let us first start the answer by writing the chemical reaction. So the chemical reaction goes as follows:
\[\text{C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{2}}}\text{Cl }\!\!~\!\!\text{ }\xrightarrow{\text{alcoholic KOH}}\text{ C}{{\text{H}}_{\text{3}}}\text{C}{{\text{H}}_{\text{2}}}\text{CH = C}{{\text{H}}_{\text{2}}}\]
In the above reaction, alcoholic KOH plays the role of a dehydrohalogenation agent. The alcoholic KOH reacts with an alkyl halide and an alkene is formed as the product of the chemical reaction.
The product that is formed consists of 4 carbon atoms and as we can see there is a double bond that is present between the first and the second carbon atoms. So, the name of the compound will be 1-butene.
The mechanism is as shown below:

The answer to the question which is 1-butene is an organic compound that is highly flammable and is a highly condensed gas. 1-butene is soluble in ether and alcohol but is not soluble in water.
So, the correct answer is “Option A”.
Additional Information:
By alcoholic solution, as mentioned in the answer, we mean a solution which is a mixture of water and ethanol, which is used as a solvent. Substances that contain sugar can be fermented using this alcoholic solution.
Note: Alcoholic potash that is used in the chemical reaction mentioned above is a very strong base. Alcoholic potash or as we know KOH dissolves in water to give $\text{R}{{\text{O}}^{-}}$ ions. Alcohol abstracts the hydrogen which results in the formation of elimination in the reaction.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




