What is the ionic bond formation of calcium and sulphur?
Answer
547.5k+ views
Hint : Factors which favour the formation of ionic bond are ionization enthalpy, electron gain enthalpy and lattice enthalpy. The energy change occurs due to formation of anion and cation, also in packing of ions of opposite charge.
Complete Step By Step Answer:
Ionic bond is formed by the transference of one or more electrons from one atom to the other. This type of bond usually comes into existence between a metal and a nonmetal atom.
The formation of ionic bond primarily depends upon:
\[1.\] The ease of formation of positive and negative ions from their respective neutral atoms.
\[2.\] The arrangement of the positive and negative ions in the solid, that is, the lattice of the crystalline compound.
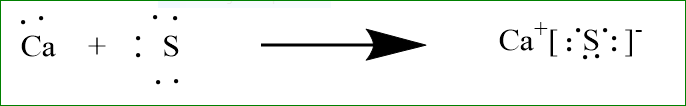
Let us study the formation of ionic bond formation of calcium and sulphur.
Sulphur has six electrons in its valence shell and it requires two electrons to attain stable gas configuration of argon. Similarly, calcium has two electrons in its valence shell and it loses two electrons to attain stable gas configuration of krypton. So, calcium transfers its valence electrons to sulphur and both form\[N{a^ + }{\text{ and }}C{l^ - }\]respectively. The electrostatic force of attraction holds these oppositely charged ions together. The above changes can be represented as:
Note :
The number of electrons which an atom loses or gains while forming an ionic bond is called its Electrovalency. The atom which loses electrons is called electropositive atom while the atom which gains an electron is called electronegative atom. Ions has a uniform field of influence around it, hence, the ionic bond is non non-directional in nature.
Complete Step By Step Answer:
Ionic bond is formed by the transference of one or more electrons from one atom to the other. This type of bond usually comes into existence between a metal and a nonmetal atom.
The formation of ionic bond primarily depends upon:
\[1.\] The ease of formation of positive and negative ions from their respective neutral atoms.
\[2.\] The arrangement of the positive and negative ions in the solid, that is, the lattice of the crystalline compound.
Let us study the formation of ionic bond formation of calcium and sulphur.
Sulphur has six electrons in its valence shell and it requires two electrons to attain stable gas configuration of argon. Similarly, calcium has two electrons in its valence shell and it loses two electrons to attain stable gas configuration of krypton. So, calcium transfers its valence electrons to sulphur and both form\[N{a^ + }{\text{ and }}C{l^ - }\]respectively. The electrostatic force of attraction holds these oppositely charged ions together. The above changes can be represented as:

Note :
The number of electrons which an atom loses or gains while forming an ionic bond is called its Electrovalency. The atom which loses electrons is called electropositive atom while the atom which gains an electron is called electronegative atom. Ions has a uniform field of influence around it, hence, the ionic bond is non non-directional in nature.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




