In which case, the carbon-carbon bond length is the same?
(A) 2-butene
(B) Benzene
(C) 1-butene
(D) 1-propyne
Answer
618k+ views
Hint: The Carbon-Carbon bond length of single bond is 154pm, and Carbon-Carbon bond length of double bond is 134pm. The bond length of the carbon-carbon triple bond is 120 pm . Conjugation decreases the bond length of a single bond and increases the bond length of double bond.
Complete step by step answer:
To see which of these options have all the same carbon-carbon bond length, we will draw the structure of each compound.
Considering option (A), 2-butene. The structure of 2-butene is as follows:
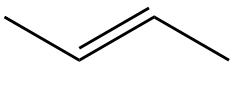
In this compound we can see that there is one double bond and two single bonds, therefore, all carbon-carbon bond lengths will not be the same. Therefore, it is not a correct option.
Considering option (B), Benzene. The structure of Benzene is as follows:
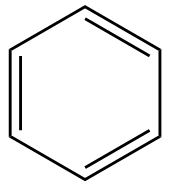
In this compound we can see that, there are 3 double bonds, there are 3 single bonds. Bond lengths are all same, instead of presence of single and bonds, because the double are in resonance, therefore, bonds are interchanged between two carbon atoms.The resonance structures of benzene is as follows:
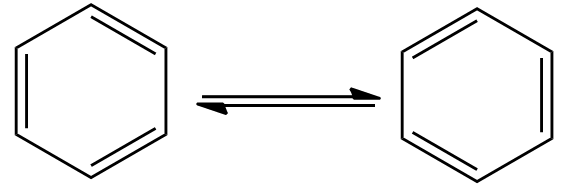
Therefore, each carbon-carbon bond is of the same length that is 140pm. Therefore, it is the correct option.
Considering option (C), 1-butene. The structure of 1-butene is as follows:

The compound 1-butene has one double bond and two single bonds therefore, the bond lengths of C-C are not equal.
Considering option (D), 1-propyne. The structure of 1-propyne is as follows:
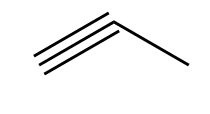
This compound has one triple bond and one single bond, so it also does not have the same C-C bond length.
So, the correct answer is “Option B”.
Note: Resonance results in partial double bond character in the molecule and hence increases the bond strength of single bonds that is the bond length after resonance decreases, and similarly decreases the bond strength of double bond and bond length increases.
Complete step by step answer:
To see which of these options have all the same carbon-carbon bond length, we will draw the structure of each compound.
Considering option (A), 2-butene. The structure of 2-butene is as follows:

In this compound we can see that there is one double bond and two single bonds, therefore, all carbon-carbon bond lengths will not be the same. Therefore, it is not a correct option.
Considering option (B), Benzene. The structure of Benzene is as follows:

In this compound we can see that, there are 3 double bonds, there are 3 single bonds. Bond lengths are all same, instead of presence of single and bonds, because the double are in resonance, therefore, bonds are interchanged between two carbon atoms.The resonance structures of benzene is as follows:

Therefore, each carbon-carbon bond is of the same length that is 140pm. Therefore, it is the correct option.
Considering option (C), 1-butene. The structure of 1-butene is as follows:

The compound 1-butene has one double bond and two single bonds therefore, the bond lengths of C-C are not equal.
Considering option (D), 1-propyne. The structure of 1-propyne is as follows:

This compound has one triple bond and one single bond, so it also does not have the same C-C bond length.
So, the correct answer is “Option B”.
Note: Resonance results in partial double bond character in the molecule and hence increases the bond strength of single bonds that is the bond length after resonance decreases, and similarly decreases the bond strength of double bond and bond length increases.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




