In Victor Meyer’s test, the colour given by ${1^0}$ , ${2^0}$ and ${3^0}$ alcohols are respectively
A) red, colourless, blue
B) red, blue, colourless
C) colourless, red, blue
D) red, blue, violet
Answer
619.5k+ views
Hint: Victor Maeyer’s test is used to differentiate primary, secondary and tertiary alcohols. In Victor Maeyer’s test, alcohols are treated with the reagents $P + {I_2}$ , $AgN{O_2}$, $HONO$ and $NaOH$ in the steps one by one. The colours obtained with the alkali helps us to distinguish the alcohols.
Complete step by step solution:
${1^0}$ , ${2^0}$ and ${3^0}$ alcohols can be distinguished using the Victor Maeyer’s test. Victor Maeyer’s test is done in four steps with the reagents $P + {I_2}$ , $AgN{O_2}$, $HONO$ and $NaOH$ respectively.
In Victor Maeyer’s test, the alcohols are treated in the following steps.
Step 1: Firstly, the sample alcohol is treated with the reagent $P + {I_2}$ to get the iodoalkane as a product.
Step 2: The iodoalkane obtained is then treated with $AgN{O_2}$ solution to get the nitroalkane.
Step 3: The nitroalkane obtained is then treated with $HONO$ (nitrous acid).
Step 4: The resulting solution in step 3 is then treated with the alkali like $NaOH$ and the colour is obtained.
Let us now do Victor Maeyer’s test of primary, secondary and tertiary alcohols.
- Victor Maeyer’s test of ${1^o}$ alcohols:
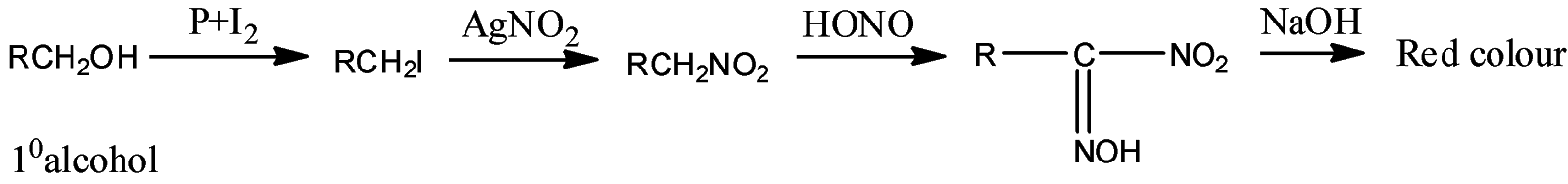
- Victor Maeyer’s test for ${2^o}$ alcohols:
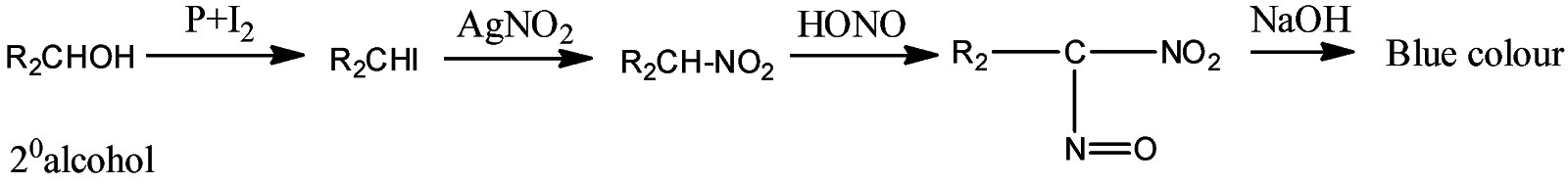
- Victor Maeyer’s test for ${3^o}$ alcohols:
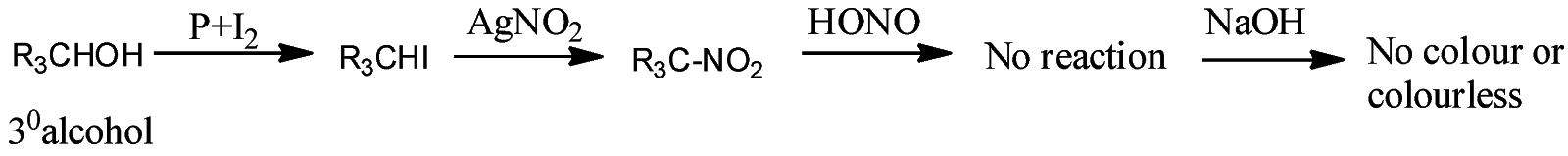
Thus, we can conclude from the above reactions of Victor Maeyer’s test of ${1^0}$, ${2^0}$ and ${3^0}$ alcohols that, primary alcohols (${1^o}$) gives the red colour solution with alkali, secondary alcohols (${2^o}$) gives the blue colour solution with alkali, and tertiary alcohols (${3^o}$) gives the colourless solution.
Thus, option (B) is the correct answer.
Note: Nitrous acid (HONO) is the combination of two reagents $NaN{O_3}$ and ${H_2}S{O_4}$. A key point to note is that Victor Maeyer’s test is not given by phenols because the procedure involves the breaking of OH bonds with carbon but in case of phenol, a carbon-oxygen bond is much stronger, having a partial bond character.
Complete step by step solution:
${1^0}$ , ${2^0}$ and ${3^0}$ alcohols can be distinguished using the Victor Maeyer’s test. Victor Maeyer’s test is done in four steps with the reagents $P + {I_2}$ , $AgN{O_2}$, $HONO$ and $NaOH$ respectively.
In Victor Maeyer’s test, the alcohols are treated in the following steps.
Step 1: Firstly, the sample alcohol is treated with the reagent $P + {I_2}$ to get the iodoalkane as a product.
Step 2: The iodoalkane obtained is then treated with $AgN{O_2}$ solution to get the nitroalkane.
Step 3: The nitroalkane obtained is then treated with $HONO$ (nitrous acid).
Step 4: The resulting solution in step 3 is then treated with the alkali like $NaOH$ and the colour is obtained.
Let us now do Victor Maeyer’s test of primary, secondary and tertiary alcohols.
- Victor Maeyer’s test of ${1^o}$ alcohols:
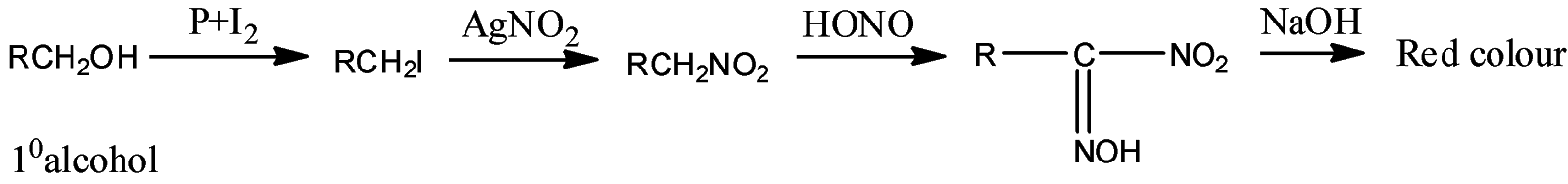
- Victor Maeyer’s test for ${2^o}$ alcohols:

- Victor Maeyer’s test for ${3^o}$ alcohols:
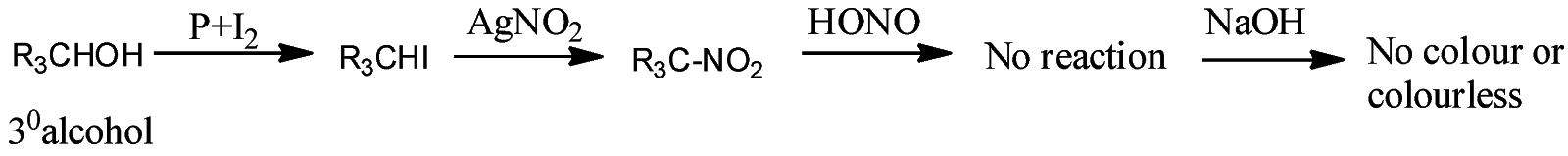
Thus, we can conclude from the above reactions of Victor Maeyer’s test of ${1^0}$, ${2^0}$ and ${3^0}$ alcohols that, primary alcohols (${1^o}$) gives the red colour solution with alkali, secondary alcohols (${2^o}$) gives the blue colour solution with alkali, and tertiary alcohols (${3^o}$) gives the colourless solution.
Thus, option (B) is the correct answer.
Note: Nitrous acid (HONO) is the combination of two reagents $NaN{O_3}$ and ${H_2}S{O_4}$. A key point to note is that Victor Maeyer’s test is not given by phenols because the procedure involves the breaking of OH bonds with carbon but in case of phenol, a carbon-oxygen bond is much stronger, having a partial bond character.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




