In thistle funnel experiment, entry of water into thistle funnel stops after sometime automatically due to
A. Diffusion of sugar out of thistle funnel.
B. External and internal solutions become isotonic.
C. Development of hydrostatic pressure in the thistle funnel.
D. Development of hydrostatic pressure in the beaker.
Answer
597k+ views
Hint: The thistle funnel experiment is widely used for the demonstration of osmosis. Osmosis is defined as a process in which molecules of a solvent or water tend to move across a semipermeable membrane from a less concentrated solution to a more concentrated solution.
Complete Answer:
- The thistle funnel experiment consists of many steps. First a thistle funnel is taken and the mouth of the funnel is tied with a goat bladder that acts as a semipermeable membrane.
- Next the thistle funnel is filled with concentrated sugar and its level in the funnel is marked. This funnel is kept in a beaker containing water. It is kept undisturbed for some time. After some time it is observed that the level of the sugar solution in the thistle funnel has increased.
- This proves that osmosis has taken place, water molecules have moved from the beaker which here is the less concentrated solution to the sugar solution which is the more concentrated solution. This process continues for some time until an equilibrium is reached after which no more osmosis takes place due to development of hydrostatic pressure in the funnel.
So the correct option is C) Development of hydrostatic pressure in the thistle funnel.
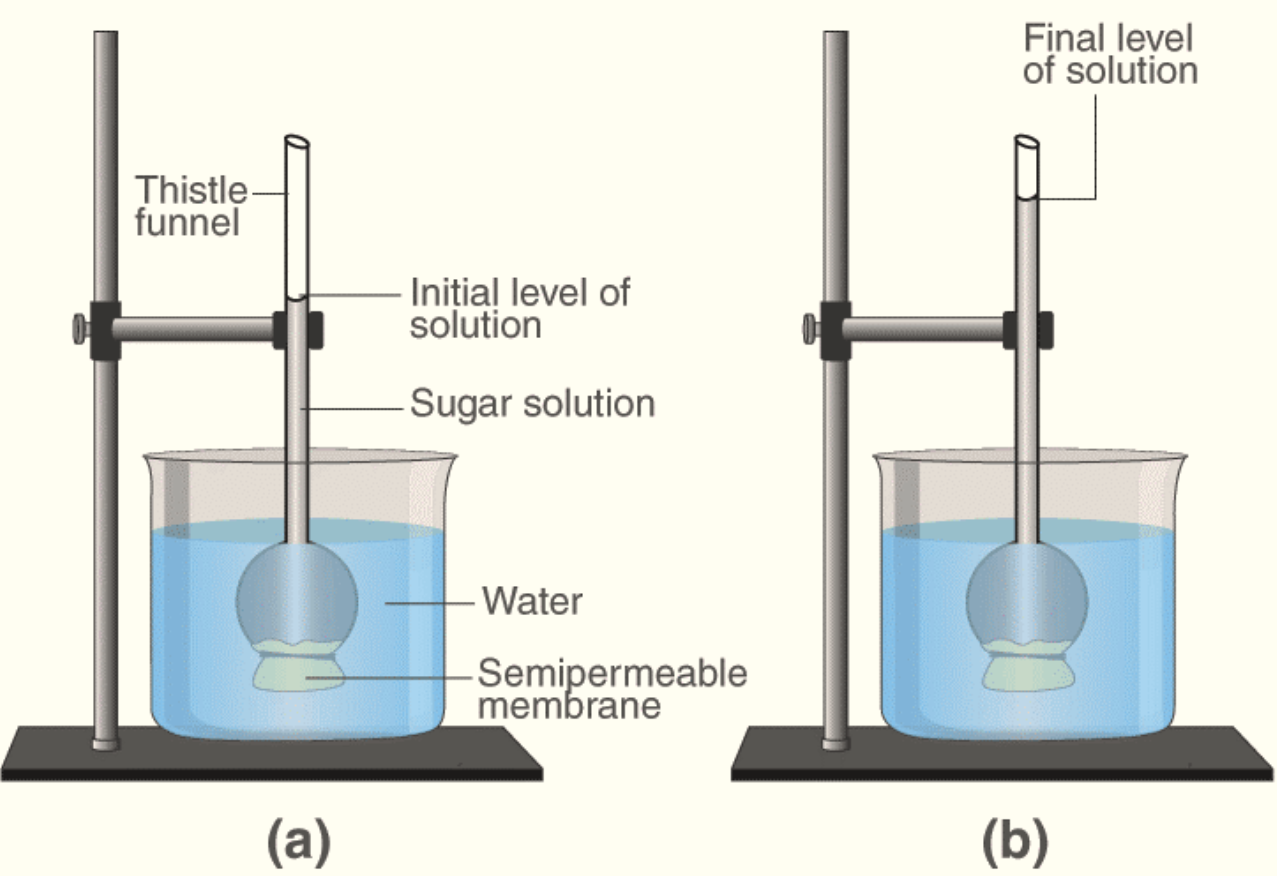
Note: Osmosis is an important process that serves various functions in both plants and animals. It’s primary role is distribution of water and nutrients to different parts.
Complete Answer:
- The thistle funnel experiment consists of many steps. First a thistle funnel is taken and the mouth of the funnel is tied with a goat bladder that acts as a semipermeable membrane.
- Next the thistle funnel is filled with concentrated sugar and its level in the funnel is marked. This funnel is kept in a beaker containing water. It is kept undisturbed for some time. After some time it is observed that the level of the sugar solution in the thistle funnel has increased.
- This proves that osmosis has taken place, water molecules have moved from the beaker which here is the less concentrated solution to the sugar solution which is the more concentrated solution. This process continues for some time until an equilibrium is reached after which no more osmosis takes place due to development of hydrostatic pressure in the funnel.
So the correct option is C) Development of hydrostatic pressure in the thistle funnel.

Note: Osmosis is an important process that serves various functions in both plants and animals. It’s primary role is distribution of water and nutrients to different parts.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




