In the reaction A is:
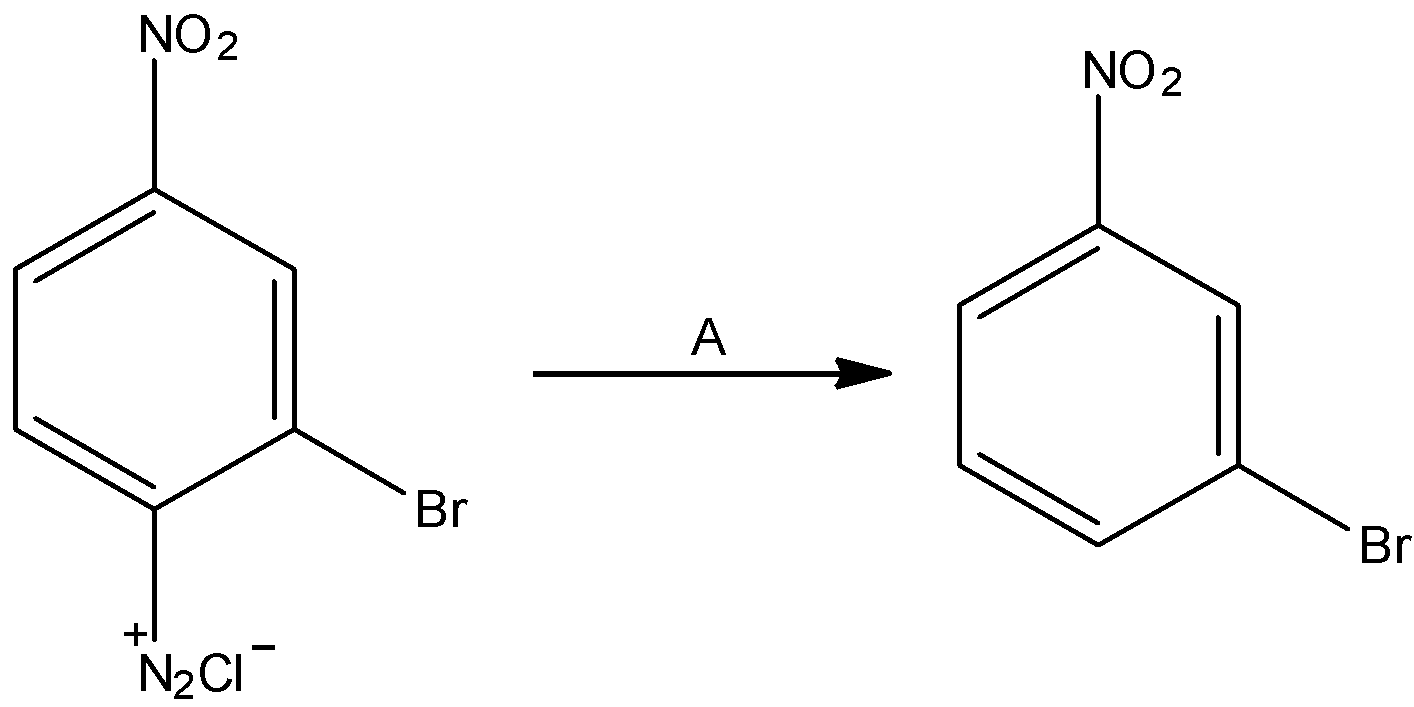
A. ${H_3}P{O_2}$
B. $C{u_2}C{l_2}$
C. $HgS{O_4}/{H_2}S{O_4}$
D. ${H^ + }/{H_2}O$

Answer
583.5k+ views
Hint: We need to know that in natural science, the reaction between arene diazonium salts with fragrant amino mixes in the acidic medium (or) reaction between arene diazonium salts with phenol in essential medium outcomes in the development of azo mixes that are splendidly hued.
Complete step by step answer:
We have to remember that the diazonium salts are integrated by the expansion of cold arrangement of sodium nitrite to arylamine arrangement in weakening corrosive at a temperature not exactly. This cycle is known as diazotization. The reaction of the benzenediazonium chloride with another compound involving a benzene ring known as coupling specialist like phenol (or) fragrant amine prompts the development of the azo compound. A few of the results of the coupling reaction are significant colors. A colored precipitate of an azo compound is delivered when the reaction of the diazonium salt with amines (or) phenols.
The given reaction is:
The diazonium group is supplanted with hydrogen molecules.
This is an advantageous strategy to eliminate an aromatic amino group in a two-step method.
The initial step is the diazotization which changes the aromatic amino group over to the diazo group and the subsequent reaction is the substitution of a diazo group by an atom of hydrogen.
In the reaction, we can see that benzene diazonium salt results in the formation of 1-bromo-3-nitrobenzene. The chemical compound is identified as ${H_3}P{O_2}$
So, the correct answer is Option A.
Note: We need to remember that the azo dyes are generally utilized in textile, fiber, cosmetic, calfskin, paint, and printing enterprises. Other than their trademark shading capacity, azo compounds are accounted for as antibacterial, antiviral, antifungal, and cytotoxic specialists. Examples of few azo compounds which are used as pH indicators are - Methyl orange, Methyl yellow, Methyl red, Congo red, Alizarin yellow. Examples of dyes that contain azo linkages areAniline yellow, Alizarin yellow R, Bismarck brown Y, and Congo red.
Complete step by step answer:
We have to remember that the diazonium salts are integrated by the expansion of cold arrangement of sodium nitrite to arylamine arrangement in weakening corrosive at a temperature not exactly. This cycle is known as diazotization. The reaction of the benzenediazonium chloride with another compound involving a benzene ring known as coupling specialist like phenol (or) fragrant amine prompts the development of the azo compound. A few of the results of the coupling reaction are significant colors. A colored precipitate of an azo compound is delivered when the reaction of the diazonium salt with amines (or) phenols.
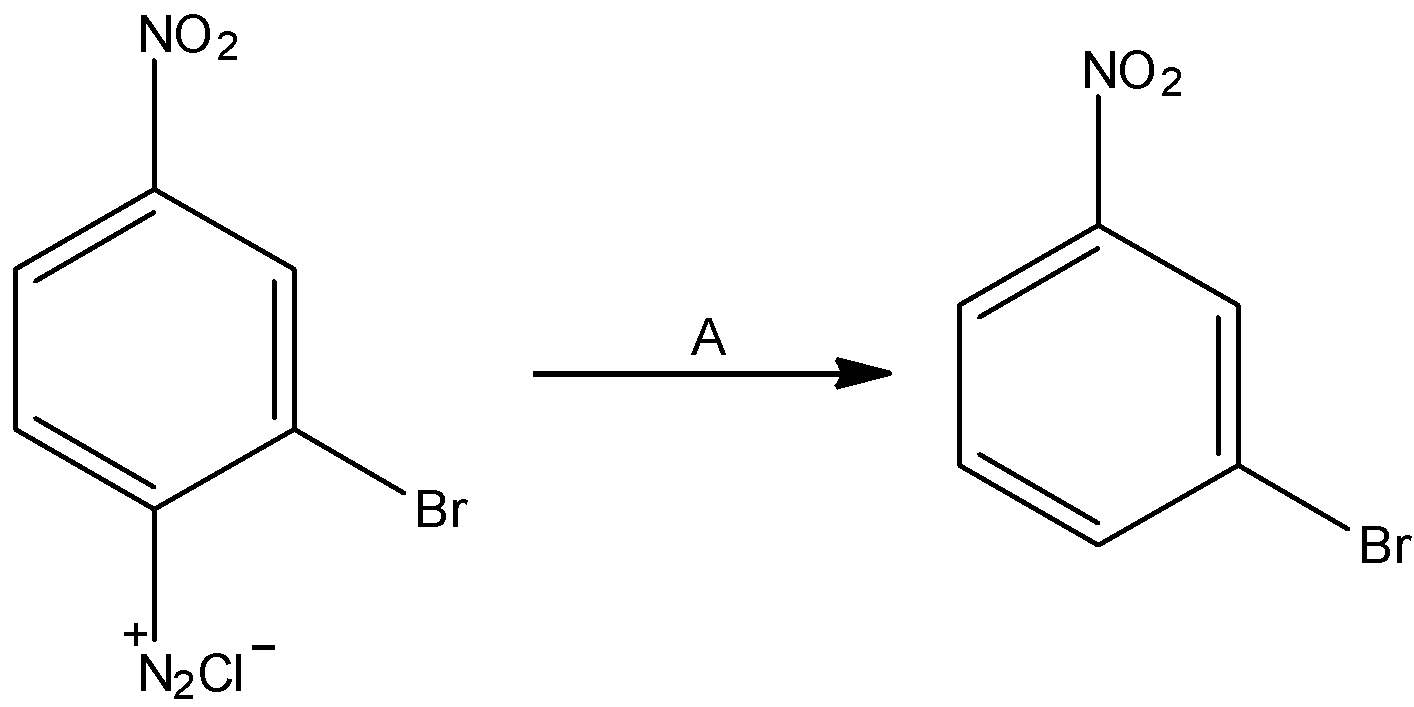
The given reaction is:

The diazonium group is supplanted with hydrogen molecules.
This is an advantageous strategy to eliminate an aromatic amino group in a two-step method.
The initial step is the diazotization which changes the aromatic amino group over to the diazo group and the subsequent reaction is the substitution of a diazo group by an atom of hydrogen.
In the reaction, we can see that benzene diazonium salt results in the formation of 1-bromo-3-nitrobenzene. The chemical compound is identified as ${H_3}P{O_2}$
So, the correct answer is Option A.
Note: We need to remember that the azo dyes are generally utilized in textile, fiber, cosmetic, calfskin, paint, and printing enterprises. Other than their trademark shading capacity, azo compounds are accounted for as antibacterial, antiviral, antifungal, and cytotoxic specialists. Examples of few azo compounds which are used as pH indicators are - Methyl orange, Methyl yellow, Methyl red, Congo red, Alizarin yellow. Examples of dyes that contain azo linkages areAniline yellow, Alizarin yellow R, Bismarck brown Y, and Congo red.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




