In the following reaction
${{(C{{H}_{3}})}_{2}}CHC{{H}_{2}}C{{H}_{3}}\xrightarrow[hv]{C{{l}_{2}}}N$ isomers of ${{C}_{5}}{{H}_{11}}Cl$.
${{C}_{5}}{{H}_{11}}Cl\xrightarrow{fractional\text{ }distillation}n$ distilled products.
The value of N and n are, respectively
A. 6, 6
B. 6, 4
C. 4, 6
D. 4, 4
Answer
603.6k+ views
Hint:. Generally alkanes undergo free radical substitution reactions with halogens in presence of sunlight. A lot of products are going to form in minor amounts (less stable) and form a major product which is a highly stable one.
Complete step by step answer:
- In the Question it is given that 2-methyl butane reacts with chlorine in presence of sunlight and forms N isomers. And on fractional distillation it forms n distilled products.
- We have to identify the N and n.
- The chemical reaction of 2-methyl butane with Chlorine in presence of sunlight is as follows.
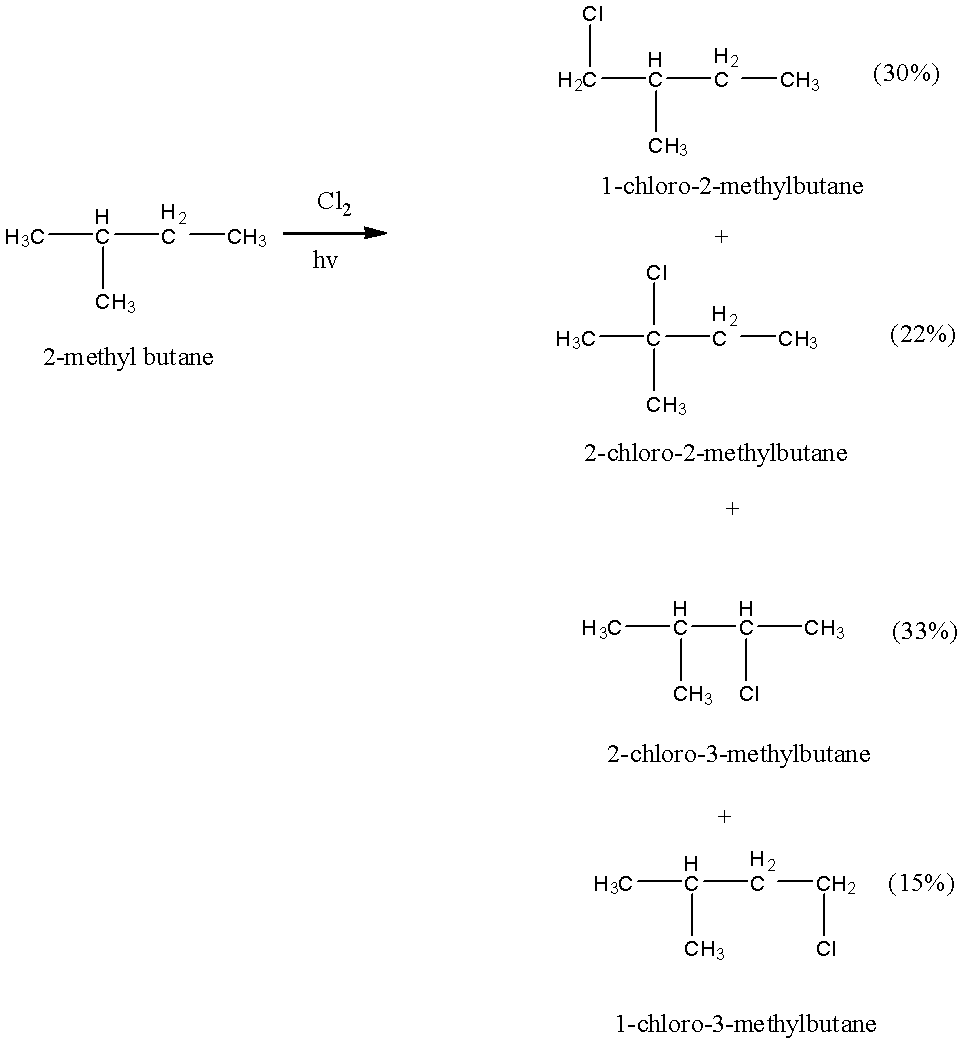
- 2-Methyl butane produces four different products with different percentages.
- The products formed are 22 % 2-chloro-2-methylbutane, 33 % 2-chloro-3-methylbutane, 30 % 1-chloro-2-methylbutane and 15 % 1-chloro-3-methylbutane.
- That means the value of N is four.
- The four products should have different boiling points due to the position of the chlorine being different from one another.
- Means on fractional distillation we will get four distilled products.
- Therefore N = 4 and n = 4.
So, the correct answer is “Option D”.
Note: By using fractional distillation we can separate the compounds on the basis of their boiling point. If a compound has less boiling point then it comes first and the compound which has a higher boiling point will come at last in the fractional distillation process.
Complete step by step answer:
- In the Question it is given that 2-methyl butane reacts with chlorine in presence of sunlight and forms N isomers. And on fractional distillation it forms n distilled products.
- We have to identify the N and n.
- The chemical reaction of 2-methyl butane with Chlorine in presence of sunlight is as follows.

- 2-Methyl butane produces four different products with different percentages.
- The products formed are 22 % 2-chloro-2-methylbutane, 33 % 2-chloro-3-methylbutane, 30 % 1-chloro-2-methylbutane and 15 % 1-chloro-3-methylbutane.
- That means the value of N is four.
- The four products should have different boiling points due to the position of the chlorine being different from one another.
- Means on fractional distillation we will get four distilled products.
- Therefore N = 4 and n = 4.
So, the correct answer is “Option D”.
Note: By using fractional distillation we can separate the compounds on the basis of their boiling point. If a compound has less boiling point then it comes first and the compound which has a higher boiling point will come at last in the fractional distillation process.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




