In Reimer-Tiemann reaction, reactants are,
A.Phenol, $CHC{l_3}$ and alkali
B.Phenol, $CC{l_4}$ and alkali
C.Aniline, $CHC{l_3}$ and alkali
D.Both Phenol, $CHC{l_3}$ and alkali and Phenol, $CC{l_4}$ and alkali
Answer
591.3k+ views
Hint: Reimer-Tiemann reaction involves ortho formylation of phenols. An aldehyde group will be introduced at the ortho position of phenol. A simple example is the conversion of phenol to salicylaldehyde.
Complete step by step answer:
Reimer-Tiemann reaction involves reaction of phenol with chloroform ($CHC{l_3}$) in the presence of an alkali, mainly NaOH. Then an aldehyde group (-CHO) will be introduced at the ortho position of the benzene ring giving the product o-hydroxybenzaldehyde or salicylaldehyde. The reaction can be represented as,
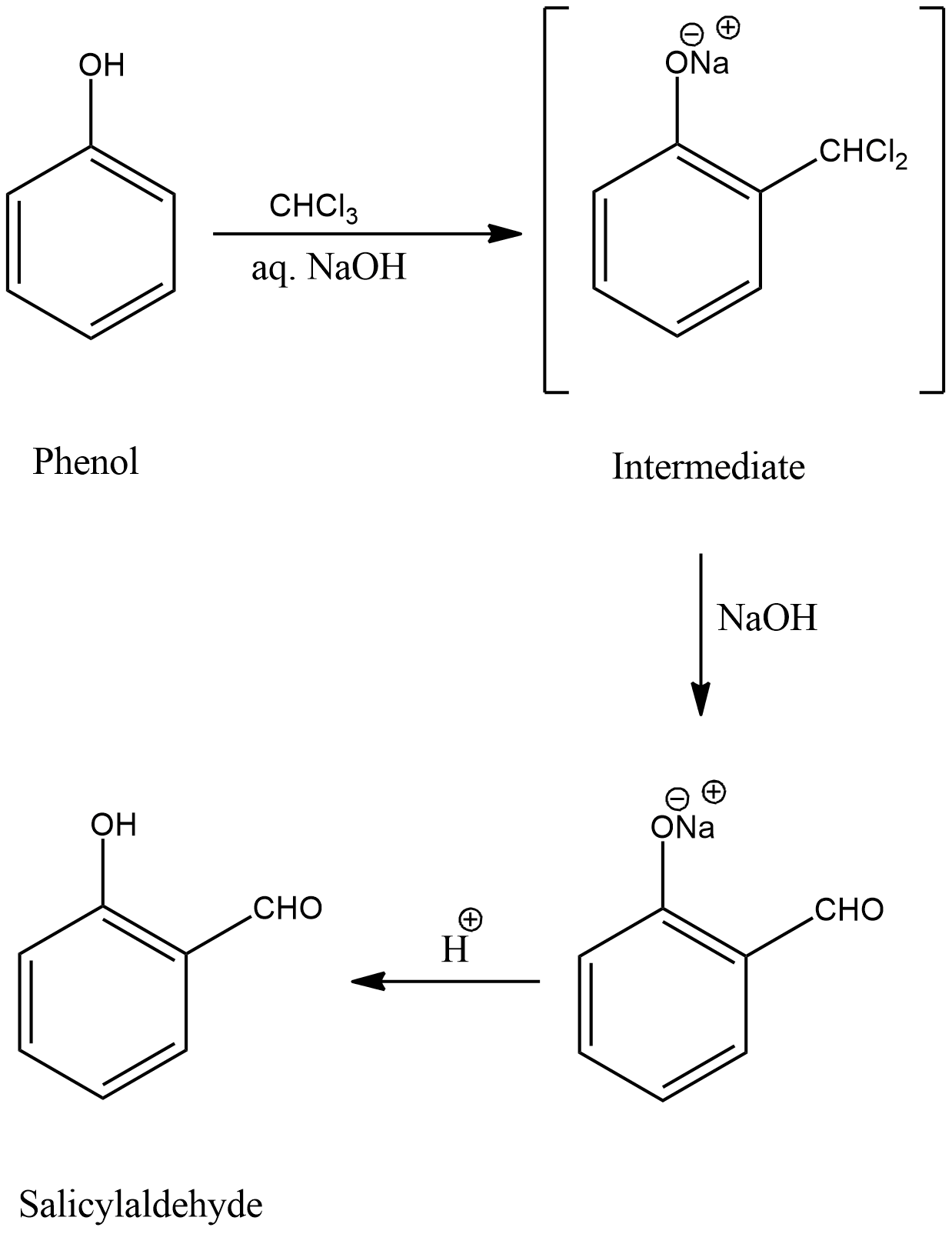
The reaction involves formation of an intermediate as shown.
We can also use $CC{l_4}$ and alkali instead of $CHC{l_3}$ and alkali as reagents. When $CC{l_4}$ and alkali is used with phenol, the product will be salicylic acid. This reaction is also a Reimer-Tiemann reaction. The corresponding reaction is shown below.
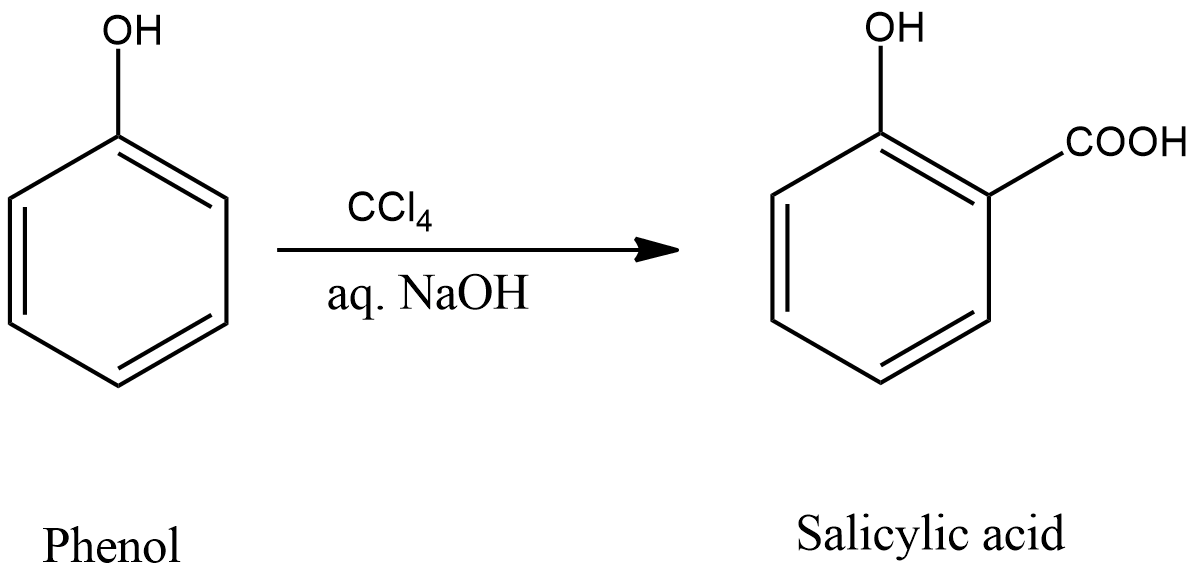
Hence both Phenol, $CHC{l_3}$ and alkali and Phenol, $CC{l_4}$ and alkali can be used in the Reimer-Tiemann reaction.
Therefore Option D is correct.
Additional information-
Hydroxides are not much soluble in chloroform. Hence in order to carry out the reaction, a biphasic solvent system should be used. The biphasic solvent system contains an aqueous hydroxide solution with an organic phase containing chloroform. During the reaction, the reagents are brought together using methods like rapid mixing, using emulsifying agents or phase transfer catalysts.
Note:
The Reimer-Tiemann reaction is more effective when other hydroxyl aromatic compounds such as naphthol are used. Heterocyclic compounds such as pyrrole and indole can also give Reimer-Tiemann reactions as they are electron rich.
Complete step by step answer:
Reimer-Tiemann reaction involves reaction of phenol with chloroform ($CHC{l_3}$) in the presence of an alkali, mainly NaOH. Then an aldehyde group (-CHO) will be introduced at the ortho position of the benzene ring giving the product o-hydroxybenzaldehyde or salicylaldehyde. The reaction can be represented as,

The reaction involves formation of an intermediate as shown.
We can also use $CC{l_4}$ and alkali instead of $CHC{l_3}$ and alkali as reagents. When $CC{l_4}$ and alkali is used with phenol, the product will be salicylic acid. This reaction is also a Reimer-Tiemann reaction. The corresponding reaction is shown below.

Hence both Phenol, $CHC{l_3}$ and alkali and Phenol, $CC{l_4}$ and alkali can be used in the Reimer-Tiemann reaction.
Therefore Option D is correct.
Additional information-
Hydroxides are not much soluble in chloroform. Hence in order to carry out the reaction, a biphasic solvent system should be used. The biphasic solvent system contains an aqueous hydroxide solution with an organic phase containing chloroform. During the reaction, the reagents are brought together using methods like rapid mixing, using emulsifying agents or phase transfer catalysts.
Note:
The Reimer-Tiemann reaction is more effective when other hydroxyl aromatic compounds such as naphthol are used. Heterocyclic compounds such as pyrrole and indole can also give Reimer-Tiemann reactions as they are electron rich.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




