In reaction,
\[Ph-NH-N{{H}_{2}}+(X)+(Y)\xrightarrow{-{{H}_{2}}O}P+Q+S\]
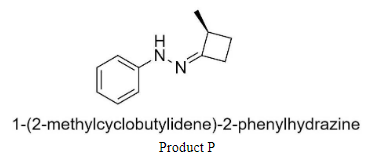
Where P is 1-(2-methylcyclo butylidene)-2-phenylhydrazine,
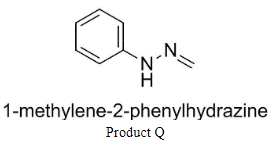
Q is 1-methylene-2-phenylhydrazine and
S is 2-methyl-4-methylene cyclobutanone.
The compounds X and Y can be?
A.
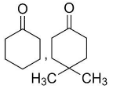
B.
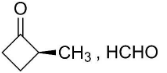
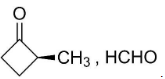
C. HCHO, $C{{H}_{3}}-CO-C{{H}_{3}}$
D.
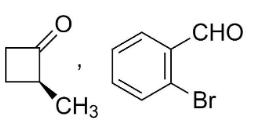

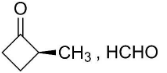

Answer
562.8k+ views
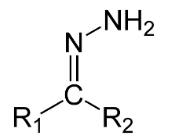
Hint: Products P and Q are examples of hydrazone compounds. Now, hydrazone is a functional group which have structural formula ${{R}_{1}}{{R}_{2}}C=NN{{H}_{2}}$. Hydrazones are formed by the addition of aldehyde or ketones to hydrazines.
Complete step-by-step answer: We know that the products P and Q are hydrazones having structures:
Now, a hydrazone is formed when the =O a functional group of an aldehyde or a ketone is replaced by $-NN{{H}_{2}}$ functional group of hydrazine. This is a type of condensation reaction which is usually accompanied by the elimination of water.
We can see that in the given question, addition of compounds X and Y to phenyl hydrazine gives products P, Q and S.
The structure of phenyl hydrazine is
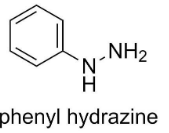
By simply looking at the structures of product P and product Q, we can determine which aldehyde or ketone is added to phenyl hydrazine to form the desired compound.
Hence to form product P, we would have to add 2-methyl-4-methylene cyclobutanone to phenyl hydrazine.
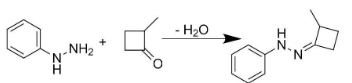
Similarly, the addition of formaldehyde to phenyl hydrazine would result in the production of compound Q.
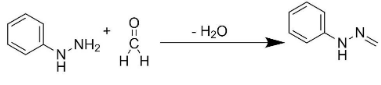
Also, the addition of X and Y would lead to the formation of product S. The entire reaction can be written as follows:
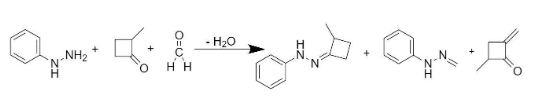
So, the compounds X and Y are option (B)
Note: Phenyl hydrazines, when used in access, also react with reducing sugars at boiling temperatures to produce hydrazones. These resultant hydrazones are known as osazones. Osazones can be used to differentiate and identify monosaccharides.

Complete step-by-step answer: We know that the products P and Q are hydrazones having structures:


Now, a hydrazone is formed when the =O a functional group of an aldehyde or a ketone is replaced by $-NN{{H}_{2}}$ functional group of hydrazine. This is a type of condensation reaction which is usually accompanied by the elimination of water.
We can see that in the given question, addition of compounds X and Y to phenyl hydrazine gives products P, Q and S.
The structure of phenyl hydrazine is

By simply looking at the structures of product P and product Q, we can determine which aldehyde or ketone is added to phenyl hydrazine to form the desired compound.
Hence to form product P, we would have to add 2-methyl-4-methylene cyclobutanone to phenyl hydrazine.

Similarly, the addition of formaldehyde to phenyl hydrazine would result in the production of compound Q.

Also, the addition of X and Y would lead to the formation of product S. The entire reaction can be written as follows:

So, the compounds X and Y are option (B)

Note: Phenyl hydrazines, when used in access, also react with reducing sugars at boiling temperatures to produce hydrazones. These resultant hydrazones are known as osazones. Osazones can be used to differentiate and identify monosaccharides.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




