In reaction between phenol and bromine water, the equivalent weight of phenol is obtained by dividing the molecular weight of phenol by:
A.3
B.4
C.5
D.6
Answer
569.7k+ views
Hint: To calculate the equivalent weight of phenol we need to write the reaction of phenol with bromine. The number of atoms of bromine required for the reaction with phenol gives a number of equivalents to be divided with.
Complete step-by-step answer:
Bromine water is basically bromine gas dissolved in water. When phenol reacts with bromine water then 3 moles of bromine reacts with one mole of phenol to form one mole of tri-substituted phenol and 3 moles of hydrogen bromide. The reaction is as follow:
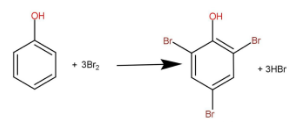
One molecule of bromine contains 2 atoms of bromines, so the number of atoms that are getting reacted are 6. So we have to divide the molecular mass of phenol with 6 to obtain the equivalent mass.
Additional information: Equivalent weight is the mass of 1 equivalent, which means it is the mass of a given substance that reacts with or combines with or displaces a fixed quantity of another substance. Equivalent weight is the ratio of molecular weight to the n factor, which is also known as valency. The definition of n-factor changes according to the type of compound. For example, in an acid n-factor is the number of protons it gives, for a salt the total number of ions it gives.
Hence the correct option is D.
Note: The n-factor in the above reaction is 6. The molecular mass of phenol is 94 and hence its equivalent weight will be \[15.6{\text{ g e}}{{\text{q}}^ - }\]. The equivalent mass will change if n factor changes for some other reaction.
Complete step-by-step answer:
Bromine water is basically bromine gas dissolved in water. When phenol reacts with bromine water then 3 moles of bromine reacts with one mole of phenol to form one mole of tri-substituted phenol and 3 moles of hydrogen bromide. The reaction is as follow:

One molecule of bromine contains 2 atoms of bromines, so the number of atoms that are getting reacted are 6. So we have to divide the molecular mass of phenol with 6 to obtain the equivalent mass.
Additional information: Equivalent weight is the mass of 1 equivalent, which means it is the mass of a given substance that reacts with or combines with or displaces a fixed quantity of another substance. Equivalent weight is the ratio of molecular weight to the n factor, which is also known as valency. The definition of n-factor changes according to the type of compound. For example, in an acid n-factor is the number of protons it gives, for a salt the total number of ions it gives.
Hence the correct option is D.
Note: The n-factor in the above reaction is 6. The molecular mass of phenol is 94 and hence its equivalent weight will be \[15.6{\text{ g e}}{{\text{q}}^ - }\]. The equivalent mass will change if n factor changes for some other reaction.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




