In electroplating an article with Nickel, anode is:
(A) An aqueous solution of nickel sulphate
(B) An article to be electroplated
(C) A block of nickel metal
(D) None of these
Answer
575.1k+ views
Hint: Nickel electroplating is a process of depositing nickel on a metal. The prepared sample is then immersed into an electrolyte solution and is used as the cathode. The nickel anode is dissolved into the electrolyte to form nickel ions. The ions travel through the solution and get deposited on the cathode.
Complete Step By Step Solution
Nickel electroplating is a technique in which a thin layer of nickel is electroplated on a metal. In this process nickel sulfate is used as an electrolyte, an article to be electroplated is placed at cathode and a block of nickel metal is used as anode. So, the correct option is (C) i.e. block of nickel metal is used as anode for the electroplating purpose.
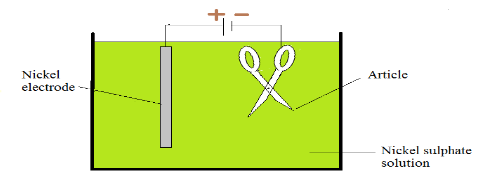
In an electrolytic cell used for electroplating, the object to be plated is used as the cathode. Metal cations are reduced at the cathode and a layer of metal is deposited on the surface of the object. The anode is usually made of the same metal as the one being plated at the cathode. A concentrated solution of ionic compounds of metal being plated is used as the electrolyte. This makes the solution very conductive.
If a nickel electrode is used as the anode, the nickel is oxidized to $ N {{i} ^ {+2}} $ ion, which goes into solution.
Reaction at anode: $N{{i}_{(s)}}\to Ni_{(aq)}^{+2}+2{{e}^{-}}$
At cathode: $Ni {aq} ^ {+2} + 2{{e} ^ {-}}\to Ni_{s} ^ {0} $
The sample gets electroplated with nickel metal.
Note
The metal with which the coating is to be done should be made anode and the article or metal at which coating is to be done should be made cathode. Parts to be plated must be clean and free of dirt, corrosion, and defects before plating can begin. Nickel is very hard and wear resistant. It is used for hard, durable, and corrosion resistant coatings.
Complete Step By Step Solution
Nickel electroplating is a technique in which a thin layer of nickel is electroplated on a metal. In this process nickel sulfate is used as an electrolyte, an article to be electroplated is placed at cathode and a block of nickel metal is used as anode. So, the correct option is (C) i.e. block of nickel metal is used as anode for the electroplating purpose.

In an electrolytic cell used for electroplating, the object to be plated is used as the cathode. Metal cations are reduced at the cathode and a layer of metal is deposited on the surface of the object. The anode is usually made of the same metal as the one being plated at the cathode. A concentrated solution of ionic compounds of metal being plated is used as the electrolyte. This makes the solution very conductive.
If a nickel electrode is used as the anode, the nickel is oxidized to $ N {{i} ^ {+2}} $ ion, which goes into solution.
Reaction at anode: $N{{i}_{(s)}}\to Ni_{(aq)}^{+2}+2{{e}^{-}}$
At cathode: $Ni {aq} ^ {+2} + 2{{e} ^ {-}}\to Ni_{s} ^ {0} $
The sample gets electroplated with nickel metal.
Note
The metal with which the coating is to be done should be made anode and the article or metal at which coating is to be done should be made cathode. Parts to be plated must be clean and free of dirt, corrosion, and defects before plating can begin. Nickel is very hard and wear resistant. It is used for hard, durable, and corrosion resistant coatings.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




