In Davisson-Germer experiment, intensity was maximum for scattering angle equal to
a) 40
b) 50
c) 60
d) 70
Answer
567.9k+ views
Hint: The scattering angle is the angle between the incident beam of electrons and the diffracted beam of electrons. This angle obtained by the experimental set up of Davisson-Germer experiment. Hence first let us understand the experimental set up and then we will see at what scattering angle is the intensity maximum.
Complete step-by-step answer:
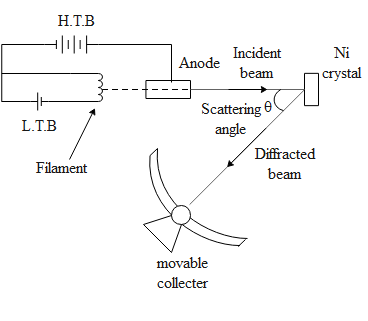
The figure above shows the set up of Davisson and Germen’s experiment.
The electrons are emitted from the hot filament of an electron gun and are accelerated using a suitable potential difference between the anode and the cathode. The fine collimated beam is directed against the face of fine Nickel crystal. The beam is further directed on to a movable detector connected to the galvanometer. Thus we measure the intensity as a function of the scattering angle , the angle between the angle between the incident and the scattered electron beam.
It was observed that there was a strong peak corresponding to a sharp diffraction maximum in the electron distribution at an accelerating voltage of 54V at and scattering angle of 50 degrees.
So, the correct answer is “Option b”.
Note: The maximum intensity obtained in the particular direction is due to constructive interference of electrons scattered from different layers of regularly spaced atoms of the crystal. The intensity is determined by the deflection in the galvanometer. It is also to be noted that the above experiment is conducted in a vacuum chamber.
Complete step-by-step answer:

The figure above shows the set up of Davisson and Germen’s experiment.
The electrons are emitted from the hot filament of an electron gun and are accelerated using a suitable potential difference between the anode and the cathode. The fine collimated beam is directed against the face of fine Nickel crystal. The beam is further directed on to a movable detector connected to the galvanometer. Thus we measure the intensity as a function of the scattering angle , the angle between the angle between the incident and the scattered electron beam.
It was observed that there was a strong peak corresponding to a sharp diffraction maximum in the electron distribution at an accelerating voltage of 54V at and scattering angle of 50 degrees.
So, the correct answer is “Option b”.
Note: The maximum intensity obtained in the particular direction is due to constructive interference of electrons scattered from different layers of regularly spaced atoms of the crystal. The intensity is determined by the deflection in the galvanometer. It is also to be noted that the above experiment is conducted in a vacuum chamber.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What is the Full Form of PVC, PET, HDPE, LDPE, PP and PS ?

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

The electric field due to a uniformly charged nonconducting class 12 physics CBSE

In a halfwave rectifier the rms value of the ac component class 12 physics CBSE

What is apomixis and what is its importance class 12 biology CBSE




