In an experiment to measure the internal resistance of a cell by a potentiometer, it is found that the balance point is at a length of 2m when the cell is shunted by a 5$\omega $ resistance and is at a length of 3m when the cell is shunted by a 10$\omega $ resistance, the internal resistance of the cell is then
$\text{A}\text{. }1.5\omega $
$\text{B}\text{. }10\omega $
$\text{C}\text{. }15\omega $
$\text{D}\text{. }1\omega $
Answer
615.9k+ views
Hint: The ratio of the potential difference across both the shunt resistances is equal to the ratio of the respective balancing lengths, i.e. $\dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{{{l}_{1}}}{{{l}_{2}}}$. The potential difference across a shunt resistance R is $V=\dfrac{ER}{(r+R)}$. Use these formulas to find the internal resistance of the cell.
Formula used:
V=iR
Complete step by step answer:
Let us first understand the working of a potentiometer. To understand better follow the given figure. This given figure is a simple setup of a potentiometer when it is used to measure the internal resistance of the unknown cell.
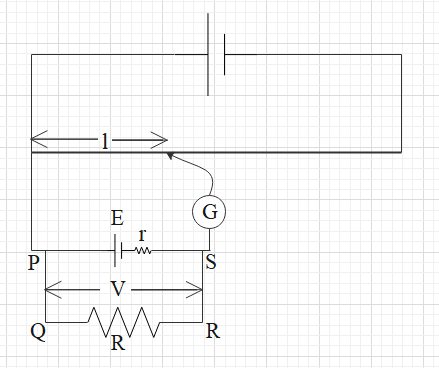
A potentiometer consists of a long wire of some known resistance and its total length is L (AB). It is connected to a cell and a potential difference is created across the wire.
J is a moveable point to find the balancing length. When there is zero current found in the galvanometer, the circuit is balanced.
When the circuit is balanced, the voltage (V) across the shunt resistance (R) is directly proportional to the balancing length (l) i.e. $V\propto l$.
Therefore, V=kl, where k is a constant.
When the circuit is balanced, $V=E-ir$ and V=iR, where I is the current in the circuit PQRS and r is the internal resistance of the cell (E).
This implies that E - ir = iR.
$\Rightarrow E=i(r+R)\Rightarrow i=\dfrac{E}{(r+R)}$.
Therefore,
$\Rightarrow V=E-ir=E-\left( \dfrac{E}{(r+R)} \right)r$
$\Rightarrow V=E\left( 1-\dfrac{r}{(r+R)} \right)=\dfrac{ER}{(r+R)}$
Let the voltage across the shunted resistance of 5$\omega $ be ${{V}_{1}}$ and the balancing length be ${{l}_{1}}$.
Therefore, ${{V}_{1}}=k{{l}_{1}}$ …. (i).
And ${{V}_{1}}=\dfrac{5E}{(r+5)}$ …. (ii).
Let the voltage across the shunted resistance of 10$\omega $ be ${{V}_{2}}$ and the balancing length be ${{l}_{2}}$.
Therefore, ${{V}_{2}}=k{{l}_{2}}$ …. (iii).
And ${{V}_{2}}=\dfrac{10E}{(r+10)}$ ….. (iv).
Divide equations (i) and (iii).
$\Rightarrow \dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{{{l}_{1}}}{{{l}_{2}}}$.
Here, ${{l}_{1}}$= 2cm and ${{l}_{2}}$= 3cm.
This gives us that
$\dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{2}{3}$.
Now, divide equations (ii) and (iv).
$\dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{\dfrac{5E}{(r+5)}}{\dfrac{10E}{(r+10)}}=\dfrac{(r+10)}{2(r+5)}$
But $\dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{2}{3}$.
Therefore,
$\dfrac{(r+10)}{2(r+5)}=\dfrac{2}{3}$
$\Rightarrow 3(r+10)=4(r+5)$
$\Rightarrow 3r+30=4r+20$
$\Rightarrow r=10\omega $
Hence, the correct option is B.
Note: Note that the emf of the unknown cell must always be less the emf of the main cell. If the emf of the cell that is to be measured is more than the emf of the main cell, then the balancing length will be more than the length of the resistance wire and we will not be able to measure the emf of the cell.
Formula used:
V=iR
Complete step by step answer:
Let us first understand the working of a potentiometer. To understand better follow the given figure. This given figure is a simple setup of a potentiometer when it is used to measure the internal resistance of the unknown cell.

A potentiometer consists of a long wire of some known resistance and its total length is L (AB). It is connected to a cell and a potential difference is created across the wire.
J is a moveable point to find the balancing length. When there is zero current found in the galvanometer, the circuit is balanced.
When the circuit is balanced, the voltage (V) across the shunt resistance (R) is directly proportional to the balancing length (l) i.e. $V\propto l$.
Therefore, V=kl, where k is a constant.
When the circuit is balanced, $V=E-ir$ and V=iR, where I is the current in the circuit PQRS and r is the internal resistance of the cell (E).
This implies that E - ir = iR.
$\Rightarrow E=i(r+R)\Rightarrow i=\dfrac{E}{(r+R)}$.
Therefore,
$\Rightarrow V=E-ir=E-\left( \dfrac{E}{(r+R)} \right)r$
$\Rightarrow V=E\left( 1-\dfrac{r}{(r+R)} \right)=\dfrac{ER}{(r+R)}$
Let the voltage across the shunted resistance of 5$\omega $ be ${{V}_{1}}$ and the balancing length be ${{l}_{1}}$.
Therefore, ${{V}_{1}}=k{{l}_{1}}$ …. (i).
And ${{V}_{1}}=\dfrac{5E}{(r+5)}$ …. (ii).
Let the voltage across the shunted resistance of 10$\omega $ be ${{V}_{2}}$ and the balancing length be ${{l}_{2}}$.
Therefore, ${{V}_{2}}=k{{l}_{2}}$ …. (iii).
And ${{V}_{2}}=\dfrac{10E}{(r+10)}$ ….. (iv).
Divide equations (i) and (iii).
$\Rightarrow \dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{{{l}_{1}}}{{{l}_{2}}}$.
Here, ${{l}_{1}}$= 2cm and ${{l}_{2}}$= 3cm.
This gives us that
$\dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{2}{3}$.
Now, divide equations (ii) and (iv).
$\dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{\dfrac{5E}{(r+5)}}{\dfrac{10E}{(r+10)}}=\dfrac{(r+10)}{2(r+5)}$
But $\dfrac{{{V}_{1}}}{{{V}_{2}}}=\dfrac{2}{3}$.
Therefore,
$\dfrac{(r+10)}{2(r+5)}=\dfrac{2}{3}$
$\Rightarrow 3(r+10)=4(r+5)$
$\Rightarrow 3r+30=4r+20$
$\Rightarrow r=10\omega $
Hence, the correct option is B.
Note: Note that the emf of the unknown cell must always be less the emf of the main cell. If the emf of the cell that is to be measured is more than the emf of the main cell, then the balancing length will be more than the length of the resistance wire and we will not be able to measure the emf of the cell.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




