In ammoniacal solution, dichromate in presence of H202 forms ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ in which \[\text{Cr}\] is in $\text{IV}$ the state. The structure is a pentagonal bipyramid. State true or false.
A) True
B) False
Answer
614.7k+ views
Hint: This ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ is a pentagonal bipyramidal structure. It involves the peroxo linkages. The chromium is tetravalent. It is synthesized from the dichromate in presence of $30{\scriptstyle{}^{0}/{}_{0}}$${{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}$.
Complete answer:
It always mentioned that only a considerable number of peroxo-chromate complexes are known. These compounds are more or less stable and these peroxo compounds decompose slowly with the evolution of oxygen. Some of these peroxo compounds are explosive.
This ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ is synthesized by the treatment of alkaline dichromate solutions $30{\scriptstyle{}^{0}/{}_{0}}$ ${{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}$. This forms the peroxo chromate$\text{(CrO}_{\text{8}}^{\text{3-}}\text{)}$. They are paramagnetic with the once unoccupied electron per formula unit.
We can say that in $\text{(CrO}_{\text{8}}^{\text{3-}}\text{)}$ the chromium is pentavalent.
When the mixture used for the preparation of ${{\text{(N}{{\text{H}}_{\text{4}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{8}}}$ is heated at the ${{50}^{0}}C$ and cooled down to ${{0}^{0}}C$ a brown crystal of ${{\text{(N}{{\text{H}}_{4}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ are obtained-ray studies has revealed the structure of ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ as shown below:
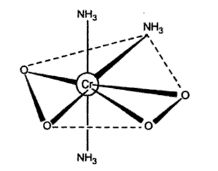
The ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ has the central metal atom as the chromium. The central chromium ion is surrounded by a total of seven ligands around it. The three are ammonia groups $\text{(N}{{\text{H}}_{\text{3}}}\text{)}$ and four of the oxygen are arranged around the central ion. The two superoxide ligands are bonded to the chromium. There are a total of five coordinating sites. For five ligands the possible structure is pentagonal bipyramidal since each oxygen of superoxide is considered as a single ligand.
Here, the oxidation state of chromium is a low valence state. The divalent chromium is coordinated to the two superoxide $\text{(-O-O-)}$ ions. The compound contains the two unpaired electrons and thus chromium is diamagnetic. In other words, we can naturally consider a compound containing the tetravalent chromium $\text{IV}$ which is coordinated peroxide ions.
Hence, when the ammoniacal solution, dichromate in presence of ${{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}$ forms ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ in which $\text{Cr}$ Is in $\text{IV}$ the state. The structure is a pentagonal bipyramid.
Hence, the statement is true. Correct option (A).
Note:
There are various peroxy –chromate for example deep blue chromium peroxide $\text{(Cr}{{\text{O}}_{\text{5}}}\text{)}$, the blue peroxy-chromate $\text{C}{{\text{r}}_{\text{2}}}\text{O}_{\text{12}}^{\text{2-}}$ , and the red peroxy-chromate $\text{(CrO}_{\text{8}}^{\text{3-}})$ .
Complete answer:
It always mentioned that only a considerable number of peroxo-chromate complexes are known. These compounds are more or less stable and these peroxo compounds decompose slowly with the evolution of oxygen. Some of these peroxo compounds are explosive.
This ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ is synthesized by the treatment of alkaline dichromate solutions $30{\scriptstyle{}^{0}/{}_{0}}$ ${{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}$. This forms the peroxo chromate$\text{(CrO}_{\text{8}}^{\text{3-}}\text{)}$. They are paramagnetic with the once unoccupied electron per formula unit.
We can say that in $\text{(CrO}_{\text{8}}^{\text{3-}}\text{)}$ the chromium is pentavalent.
When the mixture used for the preparation of ${{\text{(N}{{\text{H}}_{\text{4}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{8}}}$ is heated at the ${{50}^{0}}C$ and cooled down to ${{0}^{0}}C$ a brown crystal of ${{\text{(N}{{\text{H}}_{4}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ are obtained-ray studies has revealed the structure of ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ as shown below:

The ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ has the central metal atom as the chromium. The central chromium ion is surrounded by a total of seven ligands around it. The three are ammonia groups $\text{(N}{{\text{H}}_{\text{3}}}\text{)}$ and four of the oxygen are arranged around the central ion. The two superoxide ligands are bonded to the chromium. There are a total of five coordinating sites. For five ligands the possible structure is pentagonal bipyramidal since each oxygen of superoxide is considered as a single ligand.
Here, the oxidation state of chromium is a low valence state. The divalent chromium is coordinated to the two superoxide $\text{(-O-O-)}$ ions. The compound contains the two unpaired electrons and thus chromium is diamagnetic. In other words, we can naturally consider a compound containing the tetravalent chromium $\text{IV}$ which is coordinated peroxide ions.
Hence, when the ammoniacal solution, dichromate in presence of ${{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}$ forms ${{\text{(N}{{\text{H}}_{\text{3}}}\text{)}}_{\text{3}}}\text{Cr}{{\text{O}}_{\text{4}}}$ in which $\text{Cr}$ Is in $\text{IV}$ the state. The structure is a pentagonal bipyramid.
Hence, the statement is true. Correct option (A).
Note:
There are various peroxy –chromate for example deep blue chromium peroxide $\text{(Cr}{{\text{O}}_{\text{5}}}\text{)}$, the blue peroxy-chromate $\text{C}{{\text{r}}_{\text{2}}}\text{O}_{\text{12}}^{\text{2-}}$ , and the red peroxy-chromate $\text{(CrO}_{\text{8}}^{\text{3-}})$ .
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Name the part of the flower which the tassels of the class 12 biology CBSE

The value of constant k in coulombs law is beginalign class 12 physics CBSE

Suicide bags of cells are aEndoplasmic reticulum bLysosome class 12 biology CBSE

Quantisation of charge implies a Charge does not exist class 12 physics CBSE




