Illustrate with examples the limitations of Williamson’s synthesis for the preparation of certain types of ethers.
Answer
630.3k+ views
Hint: The reaction was developed by Alexander Williamson in 1850. A few limitations of Williamson Ether Synthesis are tertiary alkyl halides or hindered primary or secondary alkyl halides undergo elimination in the presence of an alkoxide, this nucleophile also acts as a base.
Complete step by step answer:
The Williamson ether synthesis is an organic reaction, forming an ether from a deprotonated alcohol (alkoxide) and organohalide. Reaction involves reactants as alkoxide ions with primary alkyl halide through ${{\text{S}}_{\text{N}}}2$ or bimolecular nucleophilic substitution mechanism. This is a coupling reaction.
The general reaction mechanism is-
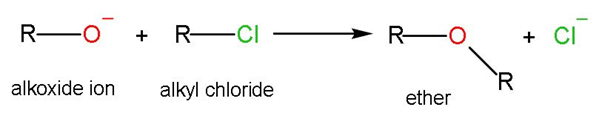
An example of this reaction is
${{\left[ {{\text{C}}_{2}}{{\text{H}}_{5}}\text{O} \right]}^{-}}{{\left[ \text{Na} \right]}^{+}}+{{\text{C}}_{2}}{{\text{H}}_{5}}\text{Cl}\to
{{\text{C}}_{2}}{{\text{H}}_{5}}\text{O}{{\text{C}}_{2}}{{\text{H}}_{5}}+\text{NaCl}$; where sodium ethoxide reacts with chloroethane to form diethyl ether.
-Limitations of Williamson’s synthesis:
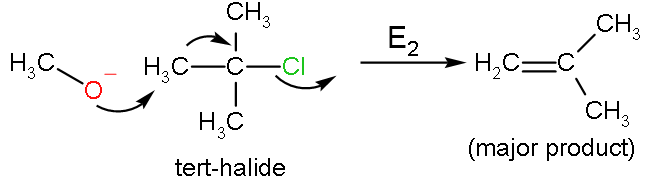
For ${{\text{S}}_{\text{N}}}2$ reaction, there must be a good leaving group which is strongly electronegative mainly a halide. In the Williamson ether reaction, there is an alkoxide ion ($\text{R}{{\text{O}}^{-}}$) which acts as the nucleophile, attacks the electrophilic carbon with leaving group, which is an alkyl tosylate. The leaving site should be a primary carbon, because secondary and tertiary carbon prefers an elimination reaction. This reaction does not favour the formation of bulky ethers such as di-tert butyl ether, due to steric hindrance and formation of alkenes is largely preferred.
Note:
The reaction between tert-butyl alcohol with primary halide is ether formation takes place, but primary alkoxide reacts with tert-butyl halide to do elimination.
Complete step by step answer:
The Williamson ether synthesis is an organic reaction, forming an ether from a deprotonated alcohol (alkoxide) and organohalide. Reaction involves reactants as alkoxide ions with primary alkyl halide through ${{\text{S}}_{\text{N}}}2$ or bimolecular nucleophilic substitution mechanism. This is a coupling reaction.
The general reaction mechanism is-

An example of this reaction is
${{\left[ {{\text{C}}_{2}}{{\text{H}}_{5}}\text{O} \right]}^{-}}{{\left[ \text{Na} \right]}^{+}}+{{\text{C}}_{2}}{{\text{H}}_{5}}\text{Cl}\to
{{\text{C}}_{2}}{{\text{H}}_{5}}\text{O}{{\text{C}}_{2}}{{\text{H}}_{5}}+\text{NaCl}$; where sodium ethoxide reacts with chloroethane to form diethyl ether.
-Limitations of Williamson’s synthesis:
For ${{\text{S}}_{\text{N}}}2$ reaction, there must be a good leaving group which is strongly electronegative mainly a halide. In the Williamson ether reaction, there is an alkoxide ion ($\text{R}{{\text{O}}^{-}}$) which acts as the nucleophile, attacks the electrophilic carbon with leaving group, which is an alkyl tosylate. The leaving site should be a primary carbon, because secondary and tertiary carbon prefers an elimination reaction. This reaction does not favour the formation of bulky ethers such as di-tert butyl ether, due to steric hindrance and formation of alkenes is largely preferred.
Note:
The reaction between tert-butyl alcohol with primary halide is ether formation takes place, but primary alkoxide reacts with tert-butyl halide to do elimination.

Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




