If in the experiment of Wheatstone’s bridge, the position of cells and galvanometer are interchanged, then the balance point will
A. change
B. remain unchanged
C. depend on the internal resistance of cell and resistance of galvanometer
D. none of these
Answer
590.4k+ views
Hint: Wheatstone bridge is an electrical circuit used to determine the resistance of an unknown resistor. It has four resistors and the ratio of two of the resistors is kept fixed. The other two resistors are balanced out of which one resistor is unknown. The unknown resistance is calculated using the null or balanced condition. So, to solve this problem, write the balanced condition of the Wheatstone bridge and then write the balanced condition after interchanging the position of cells and galvanometer. Compare these to conditions and find the answer.
Complete answer:
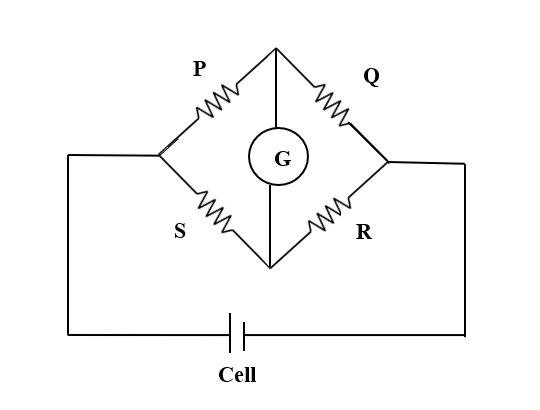
From the above figure, we can say at null condition or balanced condition of the bridge,
$\dfrac {P}{Q}= \dfrac {S}{R}$ …(1)
If we interchange the position of cell and galvanometer the circuit becomes as shown below.
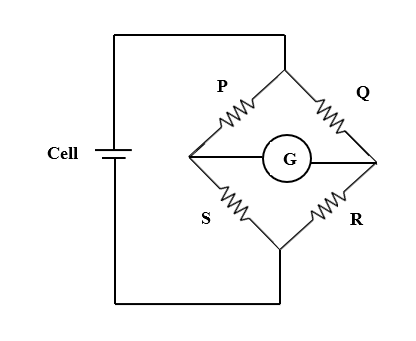
After interchanging the positions of cell and galvanometer, we can say at null condition or balanced condition of the bridge,
$\dfrac {Q}{R}= \dfrac {P}{S}$
$\Rightarrow \dfrac {S}{R}= \dfrac {P}{Q}$ …(2)
From the equation. (1) and (2) we can say before and after changing the position of cells and galvanometer, the balanced point remains unchanged.
So, the correct answer is option B i.e. remain unchanged.
Note:
Wheatstone bridge gives a very precise measurement of resistance. However, students must remember that the Wheatstone bridge is a very sensitive device and the measurements may not be precise in an off-balance condition. If all the four resistors are not comparable then the sensitivity of the circuit reduces. At the balanced condition, the current flowing through the galvanometer is zero.
Complete answer:

From the above figure, we can say at null condition or balanced condition of the bridge,
$\dfrac {P}{Q}= \dfrac {S}{R}$ …(1)
If we interchange the position of cell and galvanometer the circuit becomes as shown below.

After interchanging the positions of cell and galvanometer, we can say at null condition or balanced condition of the bridge,
$\dfrac {Q}{R}= \dfrac {P}{S}$
$\Rightarrow \dfrac {S}{R}= \dfrac {P}{Q}$ …(2)
From the equation. (1) and (2) we can say before and after changing the position of cells and galvanometer, the balanced point remains unchanged.
So, the correct answer is option B i.e. remain unchanged.
Note:
Wheatstone bridge gives a very precise measurement of resistance. However, students must remember that the Wheatstone bridge is a very sensitive device and the measurements may not be precise in an off-balance condition. If all the four resistors are not comparable then the sensitivity of the circuit reduces. At the balanced condition, the current flowing through the galvanometer is zero.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




