Identify the reagents X, Y, Z for the following products:
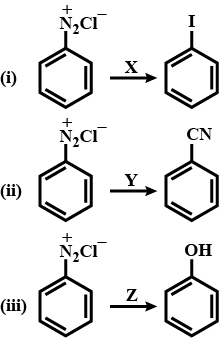
a.) X= ${ I }_{ 2 }$ warm, Y= KCN, warm, Z= NaOH, warm
b.) X= CuI, Y= NaCN, Z= KOH
c.) X= KI,warm, Y= CuCN, Z= ${ H }_{ 2 }O$,warm
d.) X=AgI,warm, Y=AgCN,warm, Z= KOH,boil

Answer
627.3k+ views
Hint: Try to recall the chemical composition of benzene diazonium chloride. Then only you can figure out why this compound is unstable and what it will form after decomposition.
Complete step by step answer:
First, let’s gain some knowledge about the species Benzenediazonium chloride.
This compound is prepared by the diazotization of aniline in the presence of hydrochloric acid. The conversion involves in situ production of nitrous acid ($HNO_{ 2 }$), which reacts with the aniline:
$C_{ 6 }H_{ 5 }NH_{ 2 }\quad +\quad HNO_{ 2 }\quad +\quad HCl\quad \rightarrow \quad [C_{ 6 }H_{ 5 }N_{ 2 }]Cl\quad +\quad 2H_{ 2 }O$
The reactions are conducted at low temperatures to minimize the decomposition of the diazonium salt. The diazonium salt is not isolated.
It is a water-soluble compound when it is at high temperature and can easily be soluble in water which explains the unstable nature. Therefore, after its preparation it was cooled down to below 5-degree centigrade and then it is stored at that temperature itself.
Benzene diazonium chloride cannot be stored and is used immediately after its preparation because of this unstable nature and with a slight increase in temperature, it dissociates to give nitrogen.
Therefore, we can conclude that option B is correct.
Note: Benzene diazonium chloride is an organic compound with the formula $ [C_{ 6 }H_{ 5 }N_{ 2 }]Cl$. It is a salt of a diazonium cation and chloride. It exists as a colorless solid that is soluble in polar solvents including water. It is the parent member of the aryl diazonium compounds, which are widely used in organic chemistry. Because the salt is unstable, it is not commercially available but is prepared upon demand.
Complete step by step answer:
First, let’s gain some knowledge about the species Benzenediazonium chloride.
This compound is prepared by the diazotization of aniline in the presence of hydrochloric acid. The conversion involves in situ production of nitrous acid ($HNO_{ 2 }$), which reacts with the aniline:
$C_{ 6 }H_{ 5 }NH_{ 2 }\quad +\quad HNO_{ 2 }\quad +\quad HCl\quad \rightarrow \quad [C_{ 6 }H_{ 5 }N_{ 2 }]Cl\quad +\quad 2H_{ 2 }O$
The reactions are conducted at low temperatures to minimize the decomposition of the diazonium salt. The diazonium salt is not isolated.
It is a water-soluble compound when it is at high temperature and can easily be soluble in water which explains the unstable nature. Therefore, after its preparation it was cooled down to below 5-degree centigrade and then it is stored at that temperature itself.
Benzene diazonium chloride cannot be stored and is used immediately after its preparation because of this unstable nature and with a slight increase in temperature, it dissociates to give nitrogen.
Therefore, we can conclude that option B is correct.
Note: Benzene diazonium chloride is an organic compound with the formula $ [C_{ 6 }H_{ 5 }N_{ 2 }]Cl$. It is a salt of a diazonium cation and chloride. It exists as a colorless solid that is soluble in polar solvents including water. It is the parent member of the aryl diazonium compounds, which are widely used in organic chemistry. Because the salt is unstable, it is not commercially available but is prepared upon demand.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




