Identify the correct option
According to $CIP$ sequence rule, the correct arrangement in order of decreasing priority is:
A. $ - OH > C{H_2}OH > - CHO > - COOH$
B. $ - OH > COOH > - CHO > - C{H_2}OH$
C. $ - COOH > OH > - COH > - C{H_2}OH$
D. $COOH > - CHO > - C{H_2}OH > - OH$
Answer
605.4k+ views
Hint:
The higher the atomic number of the immediate substituent atom, the higher the priority.
For example:
$H - < C - < N - < O - < Cl - $
Complete step by step answer:
-The $CIP$ rule is a standard process used in organic chemistry to completely and unequivocally name a stereoisomer of a molecule.
-Priority is numerically assigned as $1,2,3$ and $4$ where smaller the number higher will be the priority. So, $1$ has the highest priority and $4$ has the lowest priority.
Rules for giving the priority:
Rule$1$: Atoms having greater atomic number is given more priority
$ - OH \Rightarrow \,\,{\text{First}}$
Atomic number of ${\text{0}}$ is $8$.
Rule $2$: If ${1^{st}}$ atom is the same then, see ${2^{nd}}$ atom and apply Rule $1$.
Rule $3$: For multiple bond
I
II
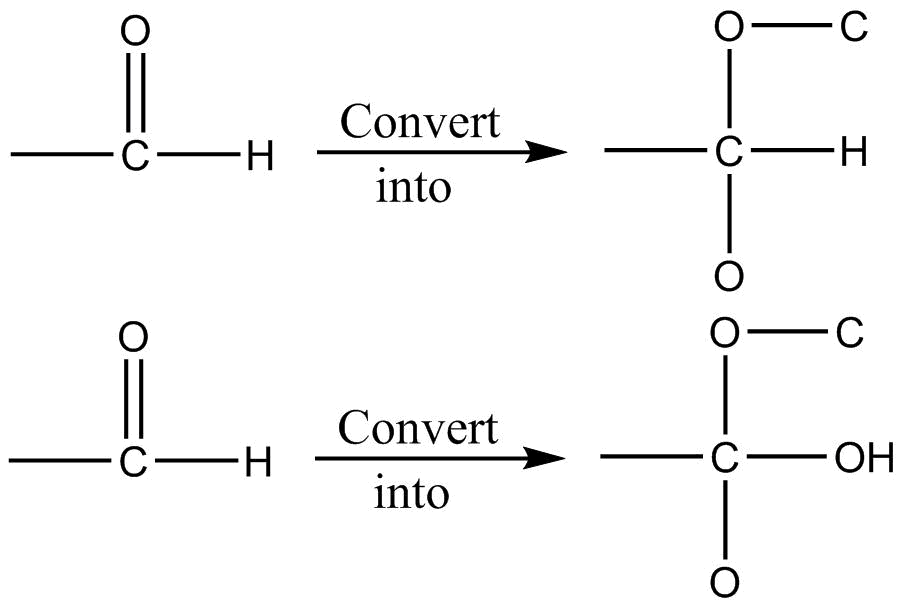
Break multiple bonds and add one more same atom to that. After this apply Rule$1$ and Rule $2$.
So, the answer is $ - OH > COOH > - CHO > - C{H_2}OH$
This is the decreasing priority order.
Multiple bonds are treated as separate single bonds.
Note: $ - CH = O$ is given priority over $ - C{H_2}OH$
$I > Br > Cl$
$OH > N{O_2} > N{H_2} > COOH > CHO > C{H_2}$
In geometric isomers:
$Z - {\text{isomer > }}E - {\text{isomer}}$
The higher the atomic number of the immediate substituent atom, the higher the priority.
For example:
$H - < C - < N - < O - < Cl - $
Complete step by step answer:
-The $CIP$ rule is a standard process used in organic chemistry to completely and unequivocally name a stereoisomer of a molecule.
-Priority is numerically assigned as $1,2,3$ and $4$ where smaller the number higher will be the priority. So, $1$ has the highest priority and $4$ has the lowest priority.
Rules for giving the priority:
Rule$1$: Atoms having greater atomic number is given more priority
$ - OH \Rightarrow \,\,{\text{First}}$
Atomic number of ${\text{0}}$ is $8$.
Rule $2$: If ${1^{st}}$ atom is the same then, see ${2^{nd}}$ atom and apply Rule $1$.
Rule $3$: For multiple bond
I
II

Break multiple bonds and add one more same atom to that. After this apply Rule$1$ and Rule $2$.
So, the answer is $ - OH > COOH > - CHO > - C{H_2}OH$
This is the decreasing priority order.
Multiple bonds are treated as separate single bonds.
Note: $ - CH = O$ is given priority over $ - C{H_2}OH$
$I > Br > Cl$
$OH > N{O_2} > N{H_2} > COOH > CHO > C{H_2}$
In geometric isomers:
$Z - {\text{isomer > }}E - {\text{isomer}}$
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




