(i) What is the role of Sulphur in the vulcanization of rubber?
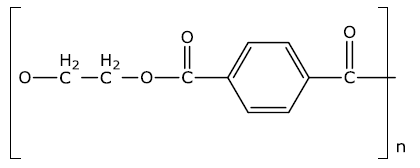
(ii) Identify the monomers in the given polymer.
(iii) Arrange the following polymers in the decreasing order of their intermolecular forces: Terylene, Polythene, Neoprene.

Answer
618.9k+ views
Hint: Mineral sulfur is generally utilized for fixing the shape of cross-joins between the elastic chains in the vulcanization procedure. During intensifying, a high versatile state of sulfur at temperatures somewhere in the range of 40°C and 70°C stretches off its particles and then breaks off these flimsy and powerless needles into pieces.
Complete answer:
Vulcanization is a compound procedure that changes over common elastic and other poldine elastomers into cross-connected polymers. The most widely recognized vulcanization operator is sulfur. Its structure spans between singular polymer atoms when warmed with elastic. Frequently an impetus and initiator are added to quicken the vulcanization procedure. The cross-connected elastomers have significantly better mechanical properties. The unvulgarized elastic has poor mechanical properties and isn't truly tough.
The monomer of the given compound is as follow:
Hexamethylenediamine - \[{{H}_{2}}N{{(C{{H}_{2}})}_{6}}N{{H}_{2}}\] and Adipic Acid -\[COOH{{(C{{H}_{2}})}_{4}}COOH\]
Decreasing order of the intermolecular forces:
Terylene > Polythene > Neoprene.
A Monomer is an iota or little particle that may tie artificially to different monomers to shape a Polymer (implies numerous parts). A Polymer is characterized as an enormous particle made out of rehashing auxiliary units.
NOTE : Monomers bind together to give shape to the polymers during a mixed response is called Polymerization, where the two particles connect together by sharing electrons.
Intermolecular Forces are the powers which act at the nuclear level. These powers keep the particles and atoms bound together. Without these powers no atom would be an acceptor. These Forces make connections between atoms, including powers among atoms and different kinds of neighboring particles.
E.g.- Atoms or particles, Hydrogen holding.
Complete answer:
Vulcanization is a compound procedure that changes over common elastic and other poldine elastomers into cross-connected polymers. The most widely recognized vulcanization operator is sulfur. Its structure spans between singular polymer atoms when warmed with elastic. Frequently an impetus and initiator are added to quicken the vulcanization procedure. The cross-connected elastomers have significantly better mechanical properties. The unvulgarized elastic has poor mechanical properties and isn't truly tough.
The monomer of the given compound is as follow:
Hexamethylenediamine - \[{{H}_{2}}N{{(C{{H}_{2}})}_{6}}N{{H}_{2}}\] and Adipic Acid -\[COOH{{(C{{H}_{2}})}_{4}}COOH\]
Decreasing order of the intermolecular forces:
Terylene > Polythene > Neoprene.
A Monomer is an iota or little particle that may tie artificially to different monomers to shape a Polymer (implies numerous parts). A Polymer is characterized as an enormous particle made out of rehashing auxiliary units.
NOTE : Monomers bind together to give shape to the polymers during a mixed response is called Polymerization, where the two particles connect together by sharing electrons.
Intermolecular Forces are the powers which act at the nuclear level. These powers keep the particles and atoms bound together. Without these powers no atom would be an acceptor. These Forces make connections between atoms, including powers among atoms and different kinds of neighboring particles.
E.g.- Atoms or particles, Hydrogen holding.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




