Grignard reagent can’t be prepared from:
A.

B.
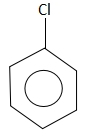
C.
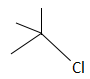
D.
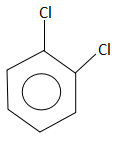




Answer
596.7k+ views
Hint: A Grignard reagent or Grignard compound is a chemical compound with the generic formula \[R - Mg - X\] , where \[X\] is a halogen and \[R\] is an organic group, normally an alkyl or aryl. Grignard compounds are popular reagents in organic synthesis for creating new carbon-carbon bonds.
Complete answer:
Grignard reagents are prepared by treating an organic halide (normally organobromine) with magnesium metal. Cyclic or acyclic ethers are required to stabilize the organomagnesium compound. Water and air, which rapidly destroy the reagent by protonolysis or oxidation, are excluded using air-free techniques. Although the reagents still need to be dry, ultrasound can allow Grignard reagents to form in wet solvents by activating the magnesium such that it consumes the water.
Grignard reagents serve as a base for protic substrates (this scheme does not show workup conditions, which typically includes water). Grignard reagents are basic and react with alcohols, phenols, etc. to give alkoxides (\[R - O - MgBr\] ).
The compound that does not take part in the formation of a Grignard reagent is:

This is so because it contains an alcoholic group which immediately reacts with the Grignard reagent. The alcoholic group is slightly acidic in nature and the hydrogen atom present on the alcohol group reacts with Grignard's reagent.
Thus, the correct option is A.
Note:
As is common for reactions involving solids and solution, the formation of Grignard reagents is often subject to an induction period. During this stage, the passivating oxide on the magnesium is removed. After this induction period, the reactions can be highly exothermic. This exothermicity must be considered when a reaction is scaled-up from laboratory to production plant. Most organohalides will work, but carbon-fluorine bonds are generally unreactive, except with specially activated magnesium.
Complete answer:
Grignard reagents are prepared by treating an organic halide (normally organobromine) with magnesium metal. Cyclic or acyclic ethers are required to stabilize the organomagnesium compound. Water and air, which rapidly destroy the reagent by protonolysis or oxidation, are excluded using air-free techniques. Although the reagents still need to be dry, ultrasound can allow Grignard reagents to form in wet solvents by activating the magnesium such that it consumes the water.
Grignard reagents serve as a base for protic substrates (this scheme does not show workup conditions, which typically includes water). Grignard reagents are basic and react with alcohols, phenols, etc. to give alkoxides (\[R - O - MgBr\] ).
The compound that does not take part in the formation of a Grignard reagent is:

This is so because it contains an alcoholic group which immediately reacts with the Grignard reagent. The alcoholic group is slightly acidic in nature and the hydrogen atom present on the alcohol group reacts with Grignard's reagent.
Thus, the correct option is A.
Note:
As is common for reactions involving solids and solution, the formation of Grignard reagents is often subject to an induction period. During this stage, the passivating oxide on the magnesium is removed. After this induction period, the reactions can be highly exothermic. This exothermicity must be considered when a reaction is scaled-up from laboratory to production plant. Most organohalides will work, but carbon-fluorine bonds are generally unreactive, except with specially activated magnesium.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




