Given below are two reactions of water with sodium and carbon dioxide, what is the nature of water in these reactions?
$\left( i \right)2Na + 2{H_2}O \to 2NaOH + {H_2}$
$\left( {ii} \right)6C{O_2} + 12{H_2}O \to {C_6}{H_{12}}{O_6} + 6{H_2}O + 6{O_2}$
A. In (ii) water acts as an oxidizing agent and in (i) it acts as a reducing agent
B. In (i) water acts as an oxidizing agent and in (ii) it acts as a reducing agent
C. In both (i) and (ii) hydrogen acts as a reducing agent.
D. In both (i) and (ii) hydrogen acts as an oxidizing agent
Answer
588.6k+ views
Hint: Oxidation is the process that occurs in an atom, ion, or molecule when there is a loss of electrons.
Oxidation number in an atom of a molecule is defined as the residual charge which an atom has or appears to have when all other atoms in a molecule or ion are removed as ions. The oxidation number in an atom may be negative, positive, or zero.
A substance which supplies oxygen or removes hydrogen is called an oxidizing agent.
A substance which supplies hydrogen and removes oxygen is called a reducing agent.
According to the electron concept, a chemical reaction in which there is a transfer of electrons from one substance (atom, ion, or molecule) to another substance is called an oxidation-reduction reaction or redox reaction.
Complete step by step answer:
The first reaction is written below:
$2Na + 2{H_2}O \to 2NaOH + {H_2}$
In the above reaction the oxidation state of hydrogen of the water molecule in the reactant part is $ + 1$ and in the product part, the oxidation state of hydrogen is 0. Therefore, we can say that reduction is taking place in the above reaction. The one which gets reduced is called an oxidizing agent and hence hydrogen of the water molecule gets reduced in this case so water will act as an oxidizing agent.
The second reaction is written below:
$6C{O_2} + 12{H_2}O \to {C_6}{H_{12}}{O_6} + 6{H_2}O + 6{O_2}$
In the above reaction, the oxidation state of oxygen of the water molecule in the reactant part is $ - 2$ and in the product part, the oxidation state of oxygen is 0. Therefore, we can say that oxidation is taking place in the above reaction. The one which gets oxidized is called a reducing agent and hence oxygen of the water molecule gets oxidized in this case so water will act as a reducing agent.
After discussing it we can conclude that in (i) water acts as an oxidizing agent and in (ii) it acts as a reducing agent.
So, the correct answer is Option B.
Note: Water was considered to be an element until 1781. However, Cavendish in 1781, showed that it can be obtained by burning hydrogen. Therefore, it is not an element. Lavoisier (1781) confirmed the compound nature of water. Davy (1800) showed that water is composed of hydrogen and oxygen in the ratio 2:1 by volume. Dumas (1842) established that water contains hydrogen and oxygen in the ratio 1:8 by weight.
The structure of the water molecule is
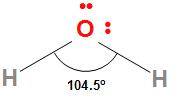
The $H - O - H$ bond angle in water is ${104.5^ \circ }$ and the $O - H$ bond length is 95.7 pm. Because of the high electronegativity difference between oxygen and hydrogen, the $O - H$ bond in water is polar. The oxygen due to high electronegativity has a partial negative charge and the hydrogen atoms due to their lowest electronegativity have partial positive charges. The dipole moment of water is 1.84 D which confirms its polar nature.
Oxidation number in an atom of a molecule is defined as the residual charge which an atom has or appears to have when all other atoms in a molecule or ion are removed as ions. The oxidation number in an atom may be negative, positive, or zero.
A substance which supplies oxygen or removes hydrogen is called an oxidizing agent.
A substance which supplies hydrogen and removes oxygen is called a reducing agent.
According to the electron concept, a chemical reaction in which there is a transfer of electrons from one substance (atom, ion, or molecule) to another substance is called an oxidation-reduction reaction or redox reaction.
Complete step by step answer:
The first reaction is written below:
$2Na + 2{H_2}O \to 2NaOH + {H_2}$
In the above reaction the oxidation state of hydrogen of the water molecule in the reactant part is $ + 1$ and in the product part, the oxidation state of hydrogen is 0. Therefore, we can say that reduction is taking place in the above reaction. The one which gets reduced is called an oxidizing agent and hence hydrogen of the water molecule gets reduced in this case so water will act as an oxidizing agent.
The second reaction is written below:
$6C{O_2} + 12{H_2}O \to {C_6}{H_{12}}{O_6} + 6{H_2}O + 6{O_2}$
In the above reaction, the oxidation state of oxygen of the water molecule in the reactant part is $ - 2$ and in the product part, the oxidation state of oxygen is 0. Therefore, we can say that oxidation is taking place in the above reaction. The one which gets oxidized is called a reducing agent and hence oxygen of the water molecule gets oxidized in this case so water will act as a reducing agent.
After discussing it we can conclude that in (i) water acts as an oxidizing agent and in (ii) it acts as a reducing agent.
So, the correct answer is Option B.
Note: Water was considered to be an element until 1781. However, Cavendish in 1781, showed that it can be obtained by burning hydrogen. Therefore, it is not an element. Lavoisier (1781) confirmed the compound nature of water. Davy (1800) showed that water is composed of hydrogen and oxygen in the ratio 2:1 by volume. Dumas (1842) established that water contains hydrogen and oxygen in the ratio 1:8 by weight.
The structure of the water molecule is

The $H - O - H$ bond angle in water is ${104.5^ \circ }$ and the $O - H$ bond length is 95.7 pm. Because of the high electronegativity difference between oxygen and hydrogen, the $O - H$ bond in water is polar. The oxygen due to high electronegativity has a partial negative charge and the hydrogen atoms due to their lowest electronegativity have partial positive charges. The dipole moment of water is 1.84 D which confirms its polar nature.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




