Give equations of the following reactions:
$ A) $ Oxidation of propan $ - 1 - $ ol with alkaline $ KMn{O_4} $ solution.
$ B) $ Bromine in $ C{S_2} $ with phenol.
$ C) $ Dilute $ HN{O_3} $ with phenol.
$ D) $ Treating phenol with chloroform in presence of aqueous $ NaOH. $
Answer
545.1k+ views
Hint: To find the products of the given reactions. We must know the type of reaction which is taking place. The type of reaction which is taking place is electrophilic substitution reaction, nitration, reimer-tiemann reaction.
Complete answer:
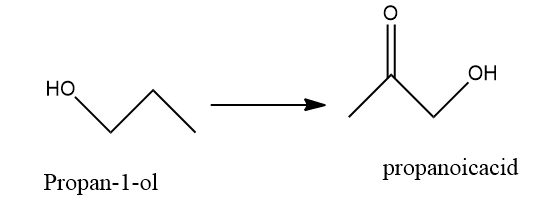
$ A) $ Oxidation of propan- $ 1 $ -ol with alkaline $ KMn{O_4} $ solution: we know that the $ KMn{O_4} $ is a strong oxidizing agent.so, it will oxidise propan- $ 1 $ -ol to propanoic acid.
$ B) $ Bromine in $ C{S_2} $ with phenol.
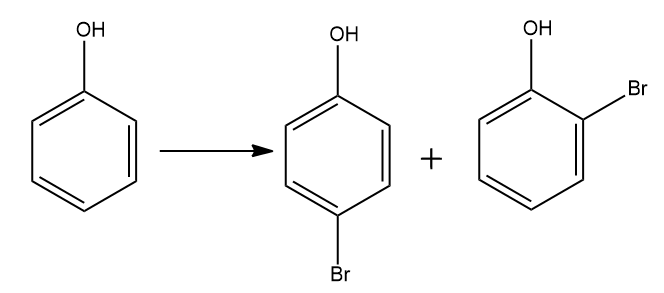
This reaction is an electrophilic substitution reaction. In an electrophilic replacement reaction, a couple of π-bonded electrons first attack an electrophile - generally a carbocation species - and a proton is then disconnected from a neighboring carbon to restore the double bond, either in the first position or with isomerization. In this reaction two products will form first is $ o - $ substituted and $ p - $ substituted.
$ C) $ Dilute $ HN{O_3} $ with phenol.
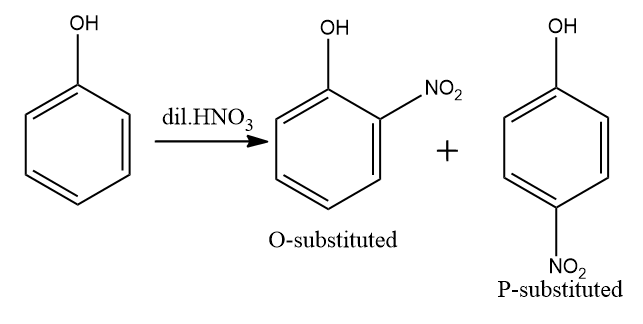
This is a nitration reaction.Nitro group into a natural compound. The term additionally is applied mistakenly to the diverse cycle of shaping nitrate esters among alcohols and nitric acid. The distinction between the subsequent subatomic designs of nitro mixtures and nitrates is that the nitrogen molecule in nitro compounds is straightforwardly attached to a non-oxygen particle, while in nitrate esters, the nitrogen is clung to an oxygen particle that thusly as a rule is clung to a carbon molecule. IN this the product will form first is $ o - $ substituted and $ p - $ substituted.
$ D) $ Treating phenol with chloroform in presence of aqueous $ HN{O_3} $ .
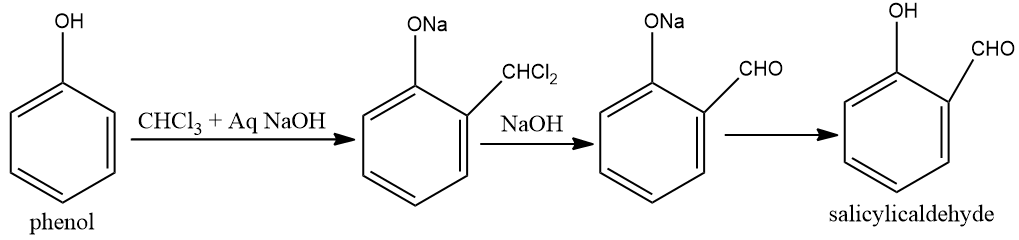
This reaction is reimer- tiemann reaction.At the point when phenols for example phenol is treated with chloroform within the sight of sodium hydroxide, an aldehyde bunch is presented at the ortho position of benzene ring prompting the arrangement of $ o $ -hydroxybenzaldehyde. The response is famously known as the Reimer Tiemann reaction.
Note:
Remember when the reaction takes place two products are formed, the major and the minor product. The major product is "more suited" than the minor product.
Complete answer:
$ A) $ Oxidation of propan- $ 1 $ -ol with alkaline $ KMn{O_4} $ solution: we know that the $ KMn{O_4} $ is a strong oxidizing agent.so, it will oxidise propan- $ 1 $ -ol to propanoic acid.

$ B) $ Bromine in $ C{S_2} $ with phenol.
This reaction is an electrophilic substitution reaction. In an electrophilic replacement reaction, a couple of π-bonded electrons first attack an electrophile - generally a carbocation species - and a proton is then disconnected from a neighboring carbon to restore the double bond, either in the first position or with isomerization. In this reaction two products will form first is $ o - $ substituted and $ p - $ substituted.

$ C) $ Dilute $ HN{O_3} $ with phenol.
This is a nitration reaction.Nitro group into a natural compound. The term additionally is applied mistakenly to the diverse cycle of shaping nitrate esters among alcohols and nitric acid. The distinction between the subsequent subatomic designs of nitro mixtures and nitrates is that the nitrogen molecule in nitro compounds is straightforwardly attached to a non-oxygen particle, while in nitrate esters, the nitrogen is clung to an oxygen particle that thusly as a rule is clung to a carbon molecule. IN this the product will form first is $ o - $ substituted and $ p - $ substituted.

$ D) $ Treating phenol with chloroform in presence of aqueous $ HN{O_3} $ .
This reaction is reimer- tiemann reaction.At the point when phenols for example phenol is treated with chloroform within the sight of sodium hydroxide, an aldehyde bunch is presented at the ortho position of benzene ring prompting the arrangement of $ o $ -hydroxybenzaldehyde. The response is famously known as the Reimer Tiemann reaction.

Note:
Remember when the reaction takes place two products are formed, the major and the minor product. The major product is "more suited" than the minor product.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




