Give condensed and bond line structural formulas and identify the functional group(s) present, if any, for 2-hydroxy-1,2,3-propane tricarboxylic acid.
Answer
597.3k+ views
Hint:We need to know what are condensed and bond line structural formulas and what functional groups are. Condensed formulas are the formulas in which the atoms are written in order. It is similar to a structural formula but written in a single line for the sake of convenience and to save space. On the other hand, bond line structural formula is a zigzag manner where every corner or terminal represents a methyl group. Functional groups are molecules responsible for their characteristic chemical reactions.
Complete step by step answer:
The given compound is 2-hydroxy-1,2,3-propane tricarboxylic acid.
1.Condensed formula: While complete structural formulas are written with the help of dashes which represent the covalent bonds, the condensed formula replaces the dashes by the groups attached to an atom by a subscript. Let us take the example of a simple molecule of ethane.
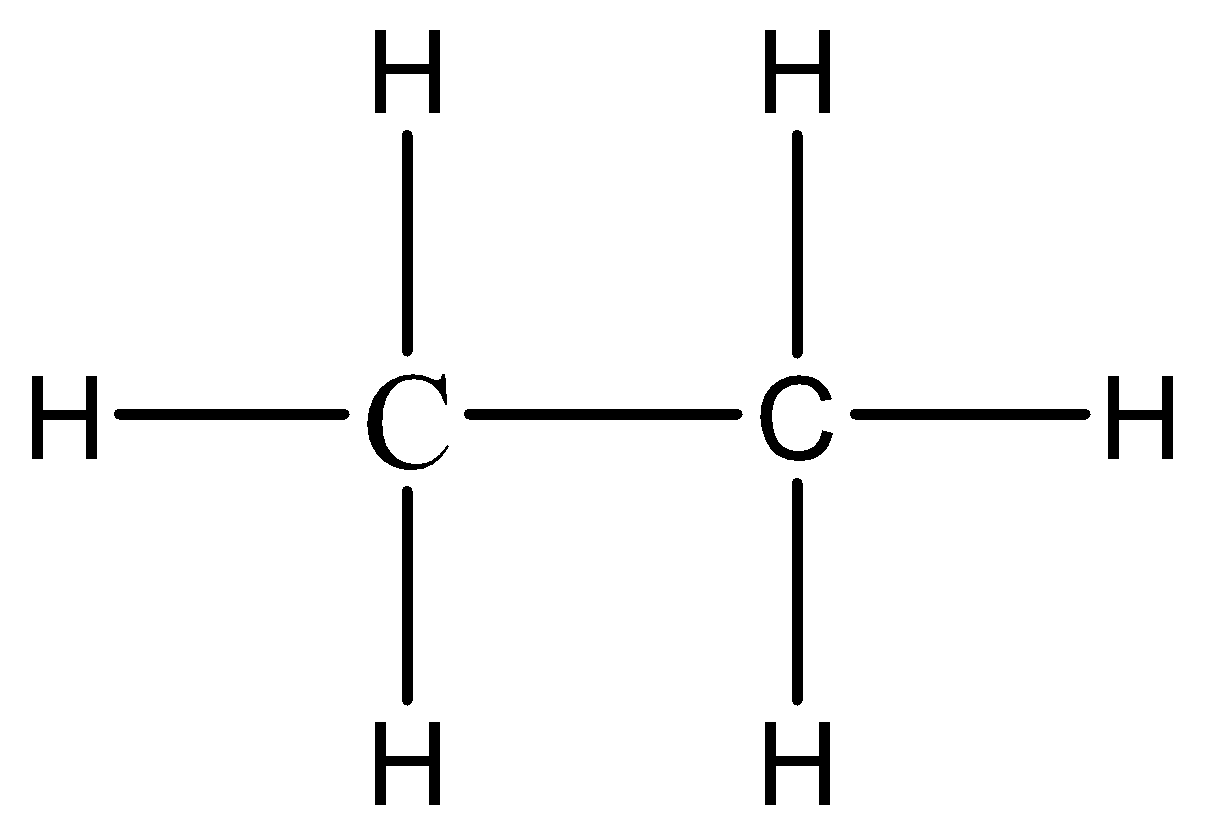
Complete structural formula of ethane.
The condensed formula of ethane will be \[C{H_3}C{H_3}\].
Therefore, the condensed formula of 2-hydroxy-1,2,3-propane tricarboxylic acid is : $\left( {COOH} \right)C{H_2}C\left( {OH} \right)\left( {COOH} \right)C{H_2}COOH$ .
2.Bond line structural formula: We must remember that the bond line structural formula is a zigzag representation where every corner represents a methyl group. For a molecule of ethane, the bond line structural formula will be a straight line with each terminal having a \[\;C{H_3}\] group.
Therefore the bond line structural formula of 2-hydroxy-1,2,3-propane tricarboxylic acid is :
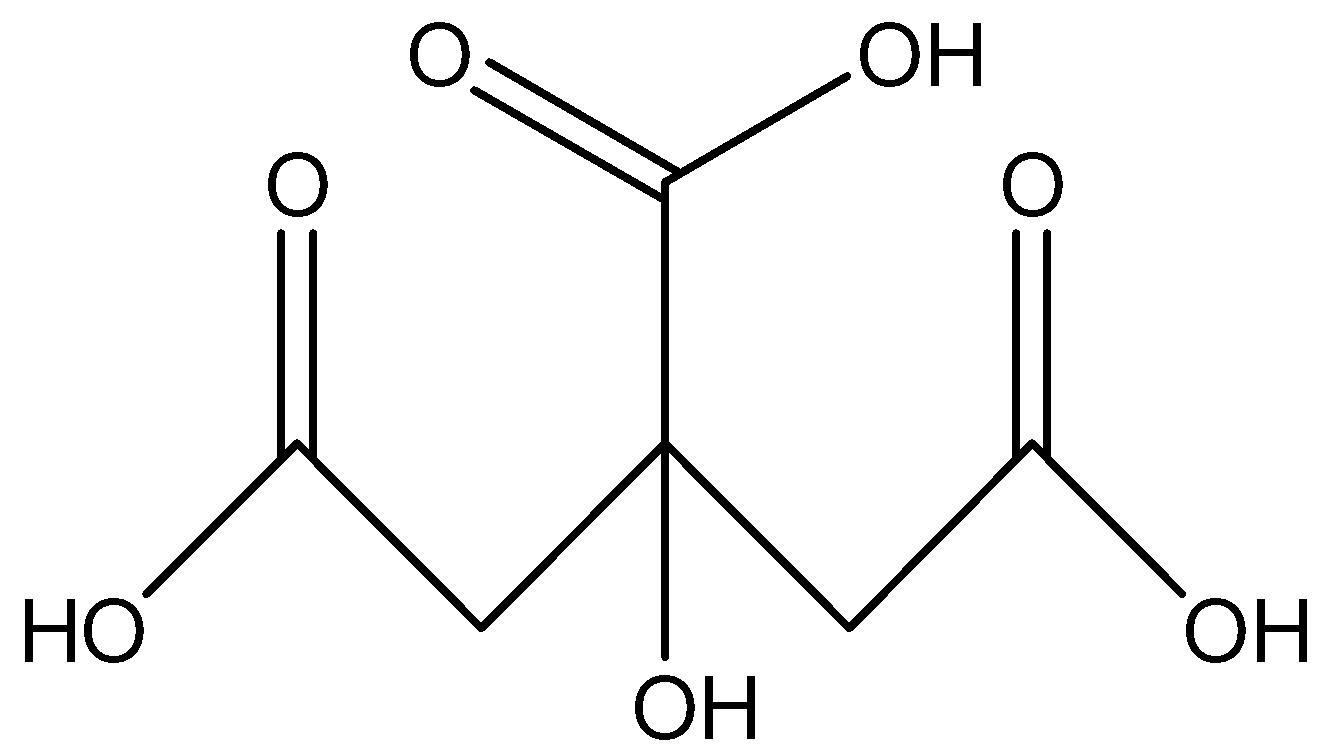
3.The functional groups present in 2-hydroxy-1,2,3-propane tricarboxylic acid are carboxylic acid ( \[ - COOH\] ) and alcoholic ( \[ - OH\] ) groups.
Note:
We have to note that these types of formulas are applicable to organic compounds only. They have only one chemical formula but can be structurally depicted in a number of ways. The bond line structural formula is also sometimes known as skeletal structure or skeletal formula. Also the molecular formula and condensed formula must not be confused. This can be explained with the example of ethane molecule whose molecular formula is represented as \[\;{C_2}{H_6}\] but the condensed formula is represented as \[C{H_3}C{H_3}\] .
Complete step by step answer:
The given compound is 2-hydroxy-1,2,3-propane tricarboxylic acid.
1.Condensed formula: While complete structural formulas are written with the help of dashes which represent the covalent bonds, the condensed formula replaces the dashes by the groups attached to an atom by a subscript. Let us take the example of a simple molecule of ethane.

Complete structural formula of ethane.
The condensed formula of ethane will be \[C{H_3}C{H_3}\].
Therefore, the condensed formula of 2-hydroxy-1,2,3-propane tricarboxylic acid is : $\left( {COOH} \right)C{H_2}C\left( {OH} \right)\left( {COOH} \right)C{H_2}COOH$ .
2.Bond line structural formula: We must remember that the bond line structural formula is a zigzag representation where every corner represents a methyl group. For a molecule of ethane, the bond line structural formula will be a straight line with each terminal having a \[\;C{H_3}\] group.
Therefore the bond line structural formula of 2-hydroxy-1,2,3-propane tricarboxylic acid is :

3.The functional groups present in 2-hydroxy-1,2,3-propane tricarboxylic acid are carboxylic acid ( \[ - COOH\] ) and alcoholic ( \[ - OH\] ) groups.
Note:
We have to note that these types of formulas are applicable to organic compounds only. They have only one chemical formula but can be structurally depicted in a number of ways. The bond line structural formula is also sometimes known as skeletal structure or skeletal formula. Also the molecular formula and condensed formula must not be confused. This can be explained with the example of ethane molecule whose molecular formula is represented as \[\;{C_2}{H_6}\] but the condensed formula is represented as \[C{H_3}C{H_3}\] .
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




